TRIM33 Reverses Cisplatin Resistance in Non-Small Cell Lung Cancer by Regulating the PI3K/AKT Pathway via Ubiquitination-Mediated Degradation of LPCAT1
DOI:
https://doi.org/10.14740/wjon2729Keywords:
Non-small cell lung cancer, Cisplatin resistance, LPCAT1, TRIM33, Ubiquitination, PI3K/AKT signaling pathway, GlycolysisAbstract
Background: Lung cancer is the leading cause of cancer-related deaths worldwide, with non-small cell lung cancer (NSCLC) accounting for 80–85% of cases. Cisplatin (DDP) is a first-line chemotherapy drug for NSCLC, but acquired DDP resistance severely limits therapeutic efficacy. Lysophosphatidylcholine acyltransferase 1 (LPCAT1) is involved in tumor progression, but its role in DDP resistance of NSCLC remains unclear. This study aimed to investigate the regulatory mechanism of LPCAT1 in DDP-resistant NSCLC and explore the potential role of tripartite motif-containing 33 (TRIM33) in modulating LPCAT1.
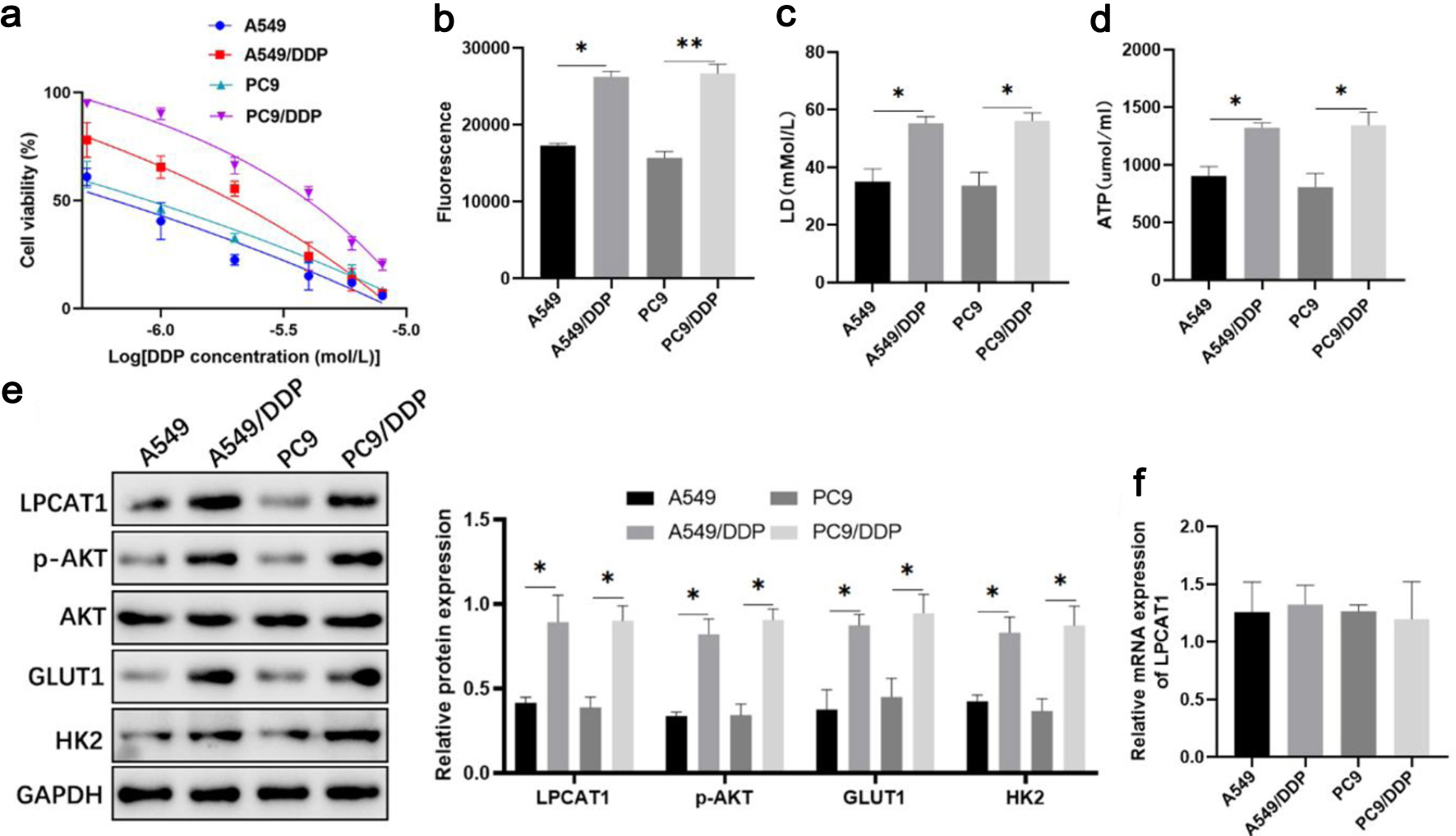
Methods: DDP-resistant NSCLC cell lines (A549/DDP, PC-9/DDP) were established by gradual concentration gradient induction. Cell viability was detected by cell counting kit-8 (CCK-8) assay to determine half-maximal inhibitory concentration (IC50) and resistance index (RI). Glucose uptake, intracellular ATP, and lactate levels were measured to evaluate glycolytic activity. Quantitative real-time polymerase chain reaction (qRT-PCR) and Western blotting were used to detect mRNA and protein expression of LPCAT1, TRIM33, and PI3K/AKT pathway-related molecules. Flow cytometry was employed to analyze cell apoptosis. Immunoprecipitation and ubiquitination assays verified the interaction between TRIM33 and LPCAT1. In vivo xenograft models were established to confirm the regulatory role of LPCAT1 and TRIM33 in DDP resistance. Statistical analyses included Student’s t-test, one-way analysis of variance (ANOVA), and Student-Newman-Keuls test, with P < 0.05 considered statistically significant.
Results: LPCAT1 protein expression was significantly upregulated in DDP-resistant NSCLC cells, while mRNA expression showed no significant difference. LPCAT1 overexpression enhanced DDP resistance, activated the PI3K/AKT signaling pathway, and promoted glycolysis in NSCLC cells, whereas LPCAT1 knockdown reversed these effects. TRIM33 expression was negatively correlated with DDP resistance, and TRIM33 directly interacted with LPCAT1 to promote its ubiquitination and degradation via the proteasomal pathway. Overexpression of TRIM33 inhibited the PI3K/AKT pathway and glycolysis, thereby sensitizing DDP-resistant cells to DDP. In vivo experiments confirmed that LPCAT1 promoted DDP resistance and tumor growth, while TRIM33 overexpression reversed this phenotype.
Conclusion: TRIM33 regulates LPCAT1 stability through ubiquitination-mediated degradation, thereby suppressing PI3K/AKT signaling and glycolysis and attenuating DDP resistance in NSCLC. These findings suggest that the TRIM33-LPCAT1 axis may represent a potential therapeutic candidate for DDP-resistant NSCLC and provide a basis for further investigation of strategies to improve DDP sensitivity.
Published
Issue
Section
License
Copyright (c) 2026 The authors

This work is licensed under a Creative Commons Attribution 4.0 International License.









