Clinical Outcomes and Prognostic Factors in Metastatic Triple-Negative Breast Cancer: A Real-World Data Analysis
DOI:
https://doi.org/10.14740/wjon2713Keywords:
Triple-negative breast cancer, Metastasis, Overall survival, Chemotherapy, Postoperative surveillanceAbstract
Background: Metastatic triple-negative breast cancer (mTNBC) is associated with poor outcomes, and therapeutic strategies remain challenging. This study analyzed real-world data to clarify clinical characteristics, treatment patterns, and survival outcomes, focusing on treatment feasibility at metastatic diagnosis.
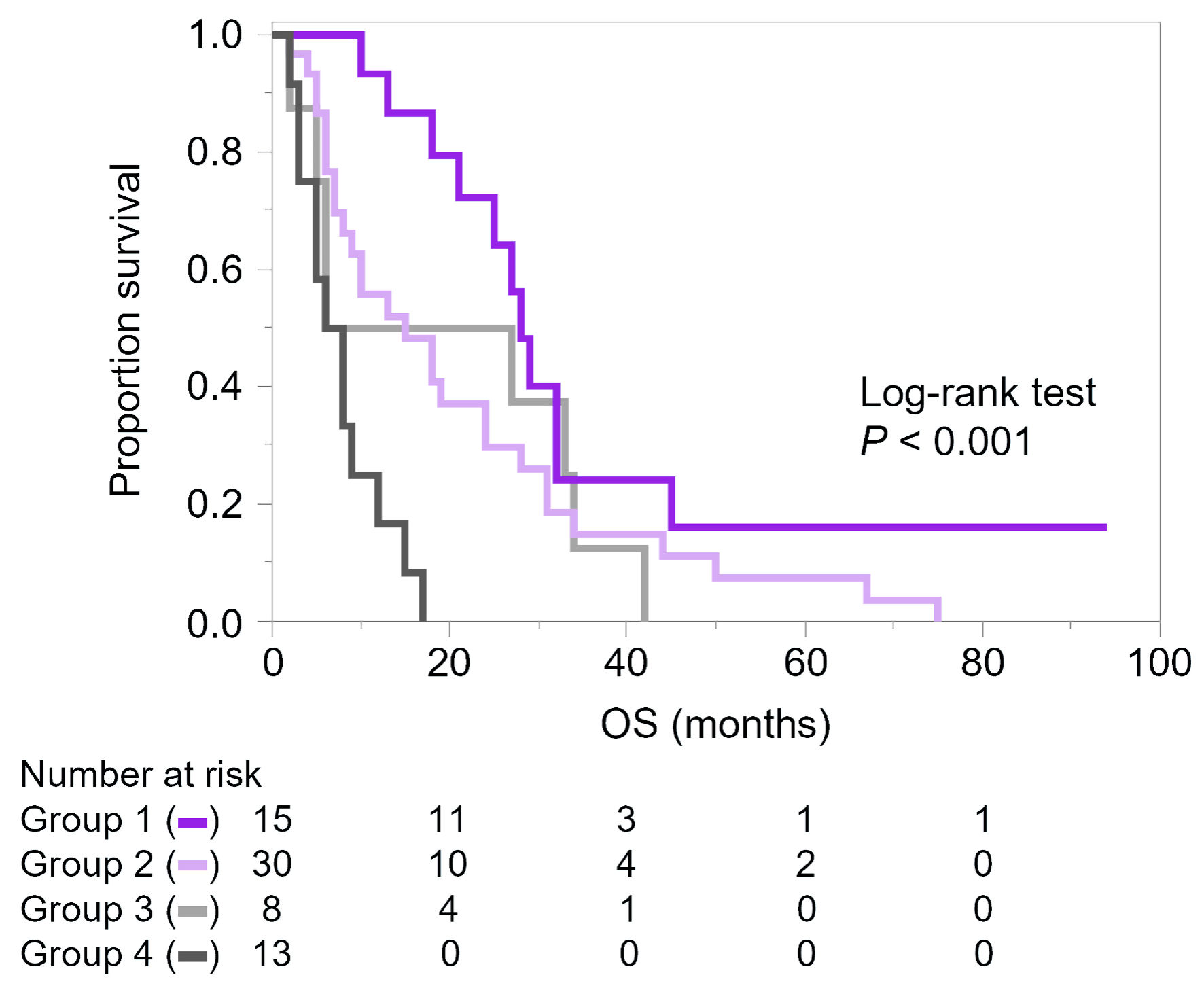
Methods: A retrospective analysis was conducted on 96 Japanese women who developed distant metastasis after curative surgery and met the inclusion criteria for the final analysis. Patients who were unable to receive drug treatment due to poor condition were defined as the non-treated group and compared with the treated group, who received at least 4 weeks of systemic therapy. Overall survival was assessed using the Cox proportional hazard model.
Results: Overall, 31% of patients could not receive systemic treatment due to poor condition. The non-treated group was more frequently diagnosed after presenting with symptoms and had a higher prevalence of poor performance status and brain metastasis (P < 0.001). Among the 66 treated patients, the median overall survival was 14 months with an average of 2.2 treatment lines. In exploratory Cox analyses, the number of metastatic organs and treatment with paclitaxel plus bevacizumab were associated with overall survival. Several patients experienced prolonged treatment with oral 5-fluorouracil.
Conclusions: A significant proportion of patients were diagnosed with mTNBC after symptom onset, limiting therapeutic intervention. Diagnosing mTNBC before symptomatic deterioration may expand treatment opportunities. Prospective evaluation of follow-up strategies and biomarkers is required, and further research should clarify treatment positioning, including paclitaxel plus bevacizumab and maintenance therapy with oral agents for biologically low-grade cases.
Published
Issue
Section
License
Copyright (c) 2026 The authors

This work is licensed under a Creative Commons Attribution 4.0 International License.









