Real-World Data on the Use of Cyclin-Dependent Kinase 4/6 Inhibitors in Hormone Receptor–Positive/Human Epidermal Growth Factor Receptor 2–Negative Advanced/Metastatic Breast Cancer
DOI:
https://doi.org/10.14740/wjon2750Keywords:
CDK4/6 inhibitors, Palbociclib, Ribociclib, Abemaciclib, Metastatic breast cancer, Real-world dataAbstract
Background: Cyclin-dependent kinase (CDK)4/6 inhibitors (CDK4/6i) have been shown to improve the outcome of patients with hormone receptor–positive (HR+) human epidermal growth factor receptor 2–negative (HER2–) advanced/metastatic breast cancer (a/mBC). This study aimed to demonstrate the safety and effectiveness of CDK4/6i in HR+/HER2- a/mBC patients, in a single-center study in Cyprus and to confirm whether these are in line with data derived from randomized clinical trials and other real-world data studies.
Methods: This retrospective study included 269 patients treated with endocrine therapy (ΕΤ) combined with palbociclib or ribociclib or abemaciclib as first-, second- or third-line treatment at the Bank of Cyprus Oncology Center (2018–2021). Seventy patients from the retrospective study who continued to receive treatment with CDK4/6i-containing regimens, were enrolled in the prospective study (follow-up period: 2021–2024) whereby patients were monitored in real time. Statistical evaluation was performed for differences in progression-free survival (PFS), overall survival (OS), adverse events (AEs)/toxicity, and prognostic factors for effectiveness.
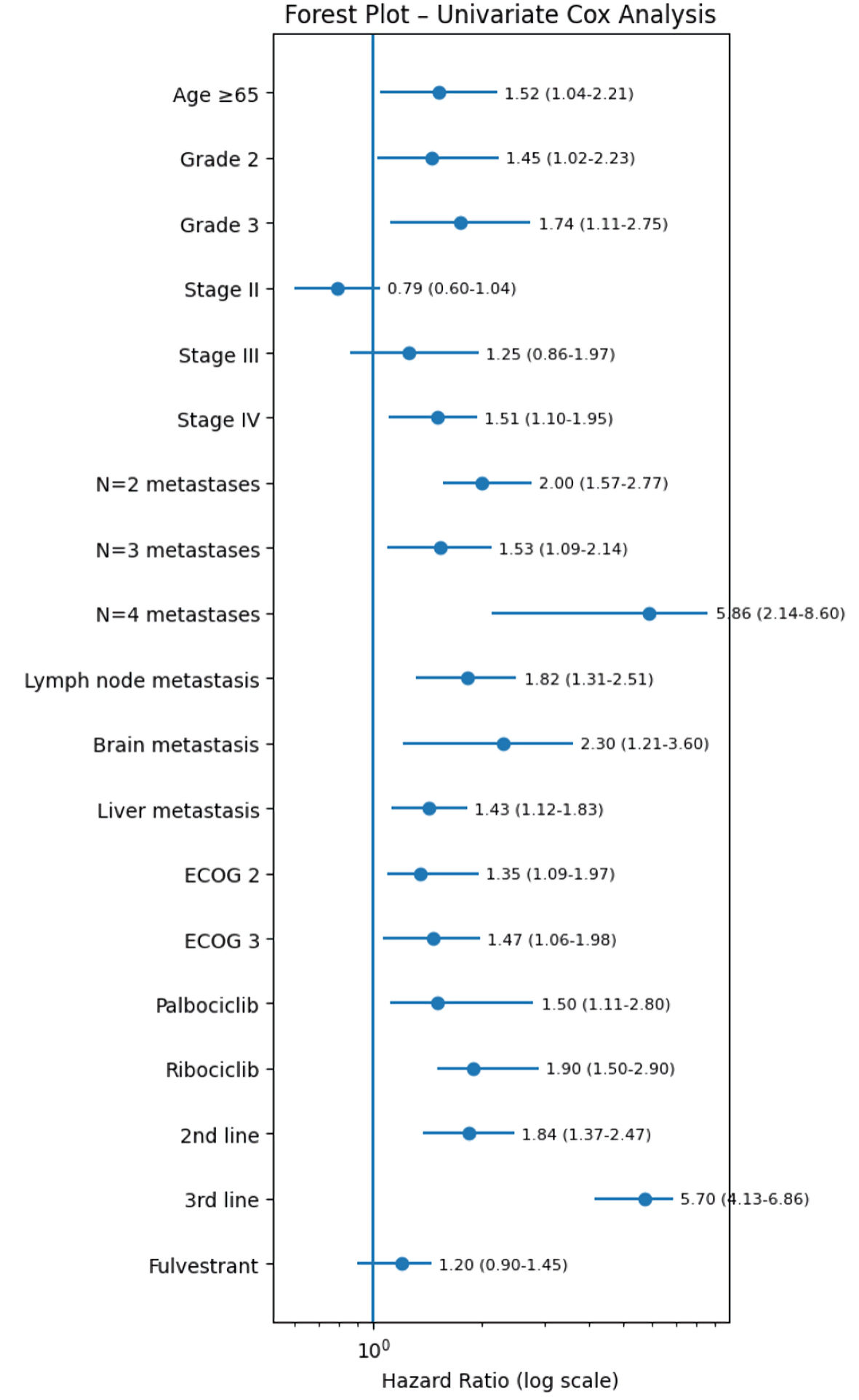
Results: The majority of patients received CDK4/6i in the first-line setting (68%, n = 182/269), whilst 25% and 7.8% of patients received CDK4/6i as second and third line of treatment, respectively. The median progression-free survival (mPFS) was 31, 25 and 19 months, with median overall survival (mOS) of 60, 54 and 44 months for palbociclib, ribociclib and abemaciclib, respectively. Neutropenia was the most commonly reported AE, followed by diarrhea, alanine aminotransferase (ALT)/aspartate aminotransferase (AST) increase, pneumonia, thrombocytopenia, erythematous rash and prolonged QT interval. Following subgroup analyses, age > 65 years, higher BC grade, de novo metastatic cancer at initial diagnosis, presence of lymph node/liver or brain metastasis and the larger number of metastatic sites at time of CDK4/6i treatment and the later-line use of CDK4/6i, were associated with a significantly shorter mPFS/mOS.
Conclusions: The combination of CDK4/6i with ET is the gold standard treatment in HR+/HER2– a/mBC. Our study results confirm the effectiveness and tolerability of CDK4/6i in clinical routine practice with prognostic factors aligning with those identified in previous studies. This is the first real–world data (RWD) describing the effectiveness and toxicity of three CDK4/6i in the Cypriot patients.
Published
Issue
Section
License
Copyright (c) 2026 The authors

This work is licensed under a Creative Commons Attribution 4.0 International License.









