Expression of Bruton’s Tyrosine Kinase Reflects Immune Cells Infiltration and Cell Proliferation in Breast Cancer
DOI:
https://doi.org/10.14740/wjon2708Keywords:
Breast cancer, BTK, Immunology, Molecular biology, Neoadjuvant chemotherapy, SurvivalAbstract
Background: Bruton’s tyrosine kinase (BTK) is a downstream mediator in B-cell receptor (BCR) signaling and is essential for B-cell differentiation and proliferation. BTK inhibitors are approved and in clinical use for hematological cancers such as lymphoma and leukemia, with testing underway in solid tumors. Because BTK is expressed in myeloid-derived suppressor cells (MDSCs) known to worsen breast cancer (BC) outcomes, we investigated the clinical relevance of BTK expression in multiple BC subtypes as a predictor of progression and/or response to treatment.
Methods: We performed an integrative transcriptomic analysis of tumor BTK expression across three large BC cohorts (The Cancer Genome Atlas (TCGA), Molecular Taxonomy of Breast Cancer International Consortium (METABRIC), and Sweden Cancerome Analysis Network-Breast (SCAN-B); total n = 5,240), 10 neoadjuvant chemotherapy (NAC) datasets, and the I-SPY2 neoadjuvant immunotherapy trial cohort. Gene set enrichment analysis (GSEA) and xCell deconvolution were used to evaluate associations with cell proliferation, immune infiltration, and tumor microenvironment (TME) composition while single-cell sequencing datasets (SCP1039, SCP1106) were used to identify BTK-expressing cell populations. Survival analyses were performed using Kaplan-Meier and log-rank tests.
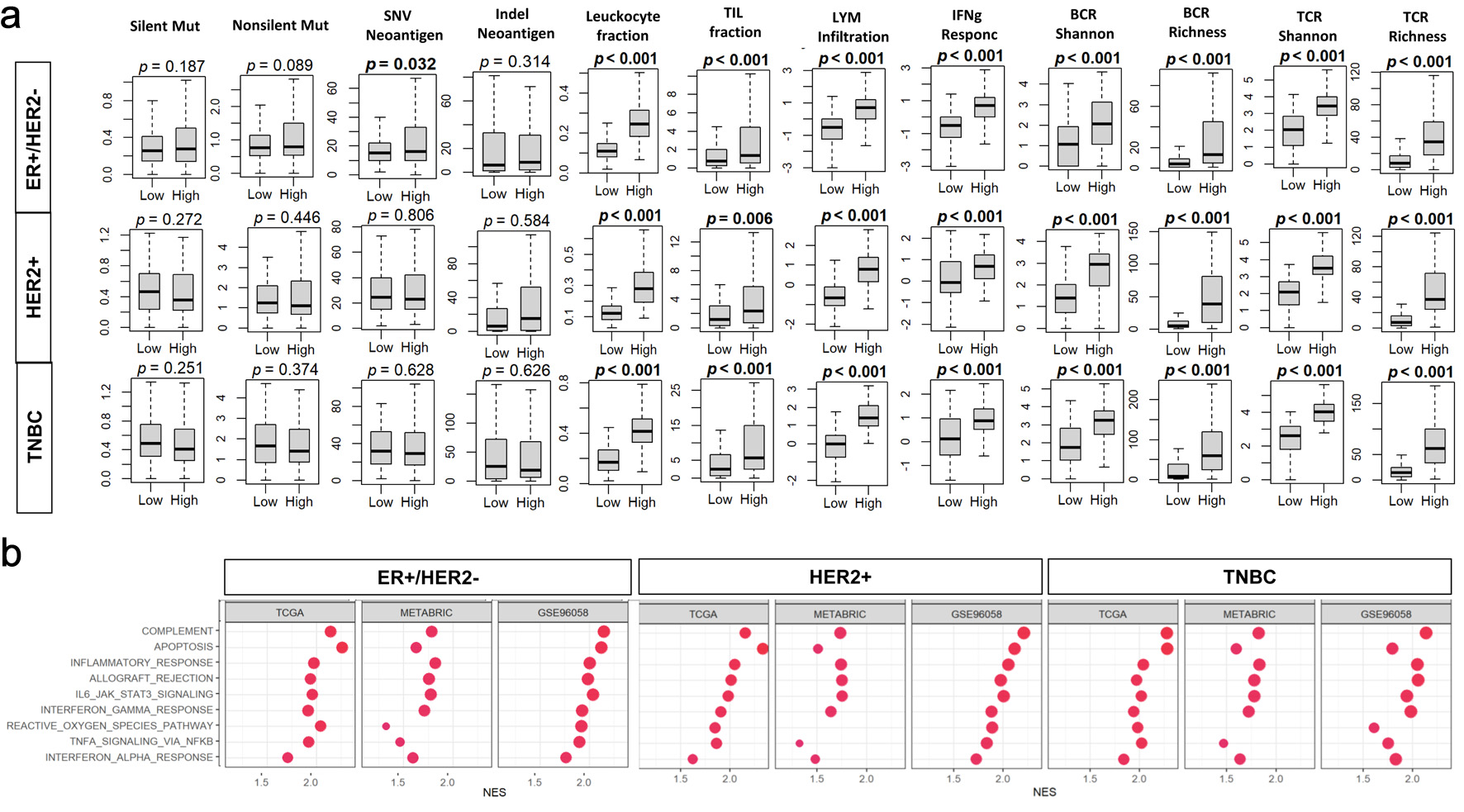
Results: BTK levels were the highest in triple-negative BC (TNBC) among the subtypes and unexpectedly drove stronger proliferation gene set enrichment in estrogen receptor-positive (ER+)/human epidermal growth factor receptor 2-negative (HER2–) tumors (e.g., MITOTIC_SPINDLE, G2M_CHECKPOINT). However, BTK expression did not correlate with American Joint Committee on Cancer (AJCC) stage or overall, disease-free, or disease-specific survival across cohorts or molecular subtypes. Notably, BTK-high tumors showed robust enrichment of immune pathways (interferon gamma (IFN-γ) response, interleukin-6 (IL-6)/Janus kinase (JAK)/signal transducer and activator of transcription 3 (STAT3), tumor necrosis factor-α (TNF-α) signaling) and exhibited elevated tumor-infiltrating leukocyte and lymphocyte fractions, increased cytolytic activity, and greater abundance of myeloid and lymphoid cell populations. BTK expression was not consistently associated with NAC response as only one of 10 datasets (GSE25066) showed a weak association within ER+ and HER2-positive subtypes. Similarly, BTK levels did not predict response in I-SPY2 patients receiving durvalumab plus olaparib, despite strong correlations with programmed cell death protein 1/programmed cell death ligand 1 (PD-1/PD-L1) expression. Single-cell analysis localized BTK transcripts primarily to myeloid and B cells.
Conclusion: BTK expression in BC reflects a proliferative and immune-active TME, particularly in TNBC and HER2-positive subtypes, but lacks prognostic or predictive value for NAC or PD-L1-based immunotherapy response.
Published
Issue
Section
License
Copyright (c) 2026 The authors

This work is licensed under a Creative Commons Attribution 4.0 International License.









