Association of Arylacetamide Deacetylase-Mediated Extracellular Matrix Remodeling With Immune Exclusion in Pancreatic Cancer
DOI:
https://doi.org/10.14740/wjon2727Keywords:
AADAC, COL6A1, Integrin signaling, CD8+ T-cell distribution, PDACAbstract
Background: Pancreatic ductal adenocarcinoma (PDAC) is a highly aggressive cancer with poor prognosis, characterized by a desmoplastic tumor microenvironment (TME) that limits immune cell infiltration and diminishes response to immunotherapy. Arylacetamide deacetylase (AADAC) is a lipid-processing enzyme, but its role in tumor progression, stromal organization, and immune modulation remains unclear.
Methods: We integrated single-cell RNA sequencing (GSE212966) and spatial transcriptomics (GSE235315) to evaluate the association of AADAC with extracellular matrix (ECM)-rich and immune-restrictive niches in PDAC. We performed spatial co-expression analysis, gene set variation analysis (GSVA) using a core COL6A1–ITGA2–ITGB1 signature, CellChat analysis of ligand–receptor interactions, and functional assays after AADAC knockdown in PANC-1 cells.
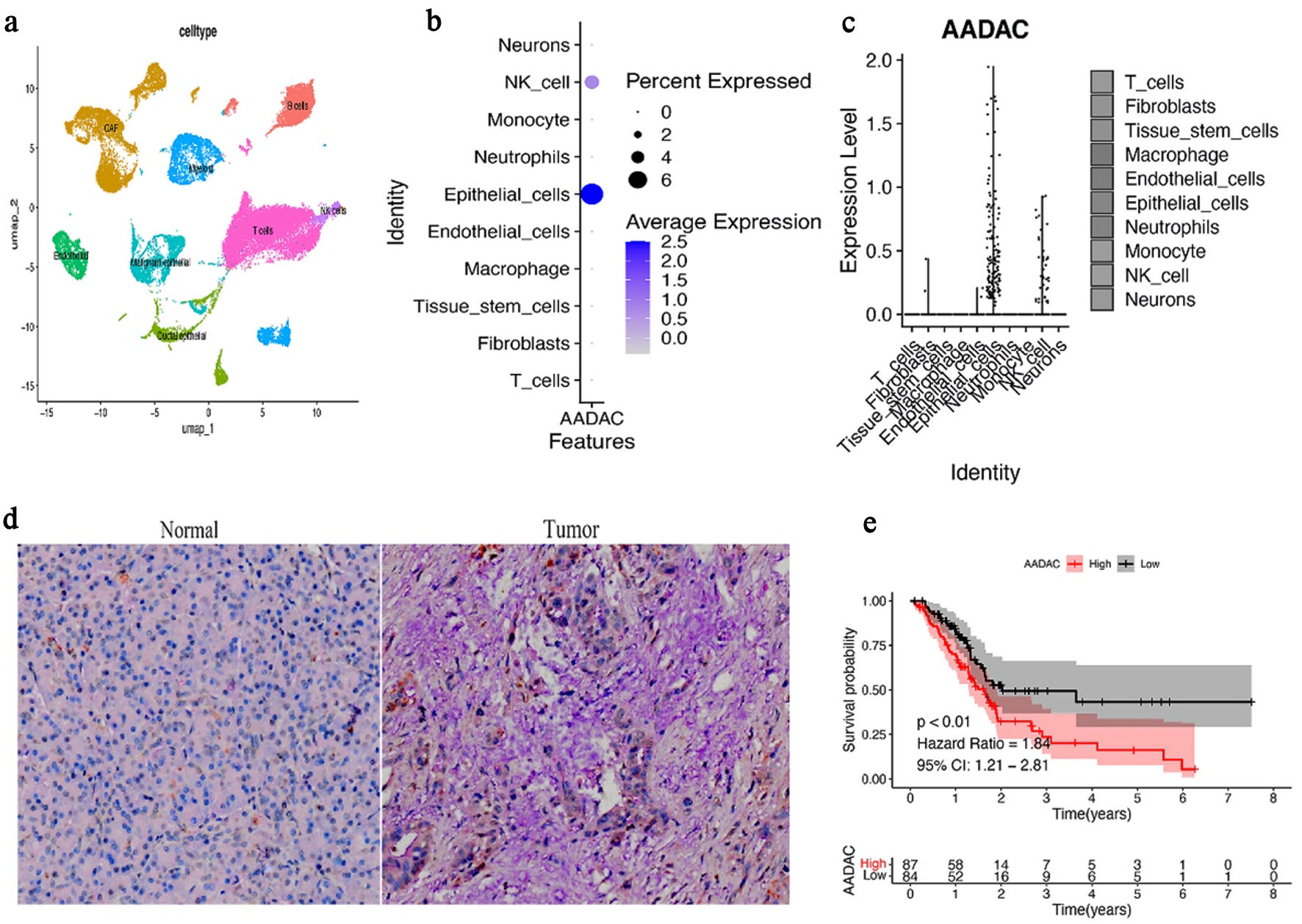
Results: AADAC expression was significantly upregulated in PDAC epithelium and co-localized with ECM markers (COL6A1, ITGA2, ITGB1), cancer-associated fibroblast (CAF)-associated markers (podoplanin (PDPN), fibroblast activation protein (FAP)), and immunosuppressive genes (TGFB1, ARG1, CD163). Spatial analyses further showed that CD8A distribution was constrained within stromalized, suppressive niches, while AADAC knockdown in PANC-1 cells increased apoptosis and reduced proliferation, migration, and invasion.
Conclusions: AADAC expression is associated with ECM-rich and immune-restrictive tumor regions in PDAC. These findings support AADAC as a candidate biomarker of epithelial–stromal–immune interactions and justify further mechanistic studies.
Published
Issue
Section
License
Copyright (c) 2026 The authors

This work is licensed under a Creative Commons Attribution 4.0 International License.









