Analysis of Prognosis and Immune Microenvironment of Protein Kinase C Substrate 80K-H in Diabetic Lung Cancer Patients
DOI:
https://doi.org/10.14740/wjon2663Keywords:
Lung cancer, Diabetes, RAGE, PRKCSH, Prognosis, Immune cell infiltration, Bioinformatics analysisAbstract
Background: A substantial association has been established between diabetes and an elevated risk of lung cancer. This study aimed to elucidate the prognosis and characterize alterations in the immune microenvironment linked to the protein kinase C substrate 80K-H (PRKCSH) gene in the context of diabetic lung cancer.
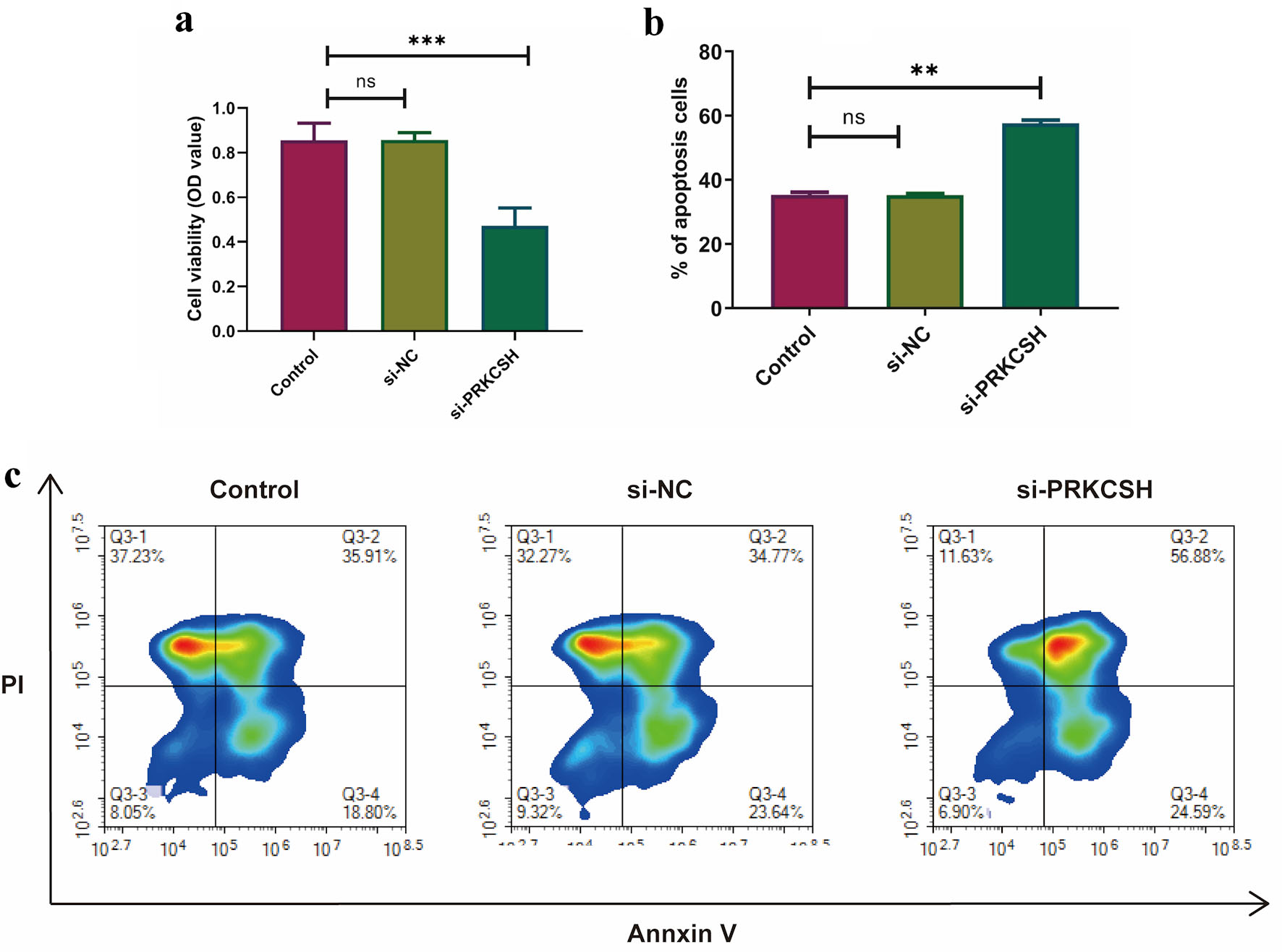
Methods: The expression profile of receptor for advanced glycation end products (RAGE) genes in lung adenocarcinoma (LUAD) and diabetic cohorts was analyzed utilizing data from The Cancer Genome Atlas (TCGA) and the Gene Expression Omnibus (GEO). The methodological framework included single-sample gene set enrichment analysis (ssGSEA), hallmark pathway enrichment analysis, Pearson’s correlation, and Wilcoxon tests, employing data from the Cancer Single Cell State Atlas (CancerSEA), tracking tumor immunophenotype (TIP) meta-server, and the Genomics of Drug Sensitivity in Cancer (GDSC) platform. PRKCSH-targeting small interfering RNA (siRNA) was synthesized and transfected into A549 cells. Functional validation of PRKCSH was conducted using real-time quantitative polymerase chain reaction (RT-qPCR), Western blotting, methylthiazolyldiphenyl-tetrazolium bromide (MTT) assays and flow cytometry.
Results: The analysis identified five RAGE genes with dysregulated expression in both diabetic and LUAD conditions, which were significantly associated with the activation of signaling pathways and patterns of immune cell enrichment in diabetes. PRKCSH has been identified as an independent prognostic marker in LUAD, with associations with key biological processes such as cell cycle regulation, genomic instability responses, inflammatory mediation, and stem cell characteristics. Comprehensive pathway analysis revealed inverse relationships between PRKCSH expression and immune-related molecular mechanisms. Detailed immune profiling indicated reduced infiltration levels of various immune cell populations in association with elevated PRKCSH expression. Notably, increased PRKCSH activity in LUAD was linked to enhanced enzymatic pathway responses and greater therapeutic sensitivity to specific enzyme inhibitors. Experimental validation via gene silencing demonstrated that suppression of PRKCSH effectively reduced malignant cell proliferation while promoting apoptotic mechanisms in lung cancer models.
Conclusions: This extensive investigation positioned PRKCSH as a critical prognostic biomarker and a promising therapeutic target for personalized immunotherapeutic strategies in the management of LUAD.
Published
Issue
Section
License
Copyright (c) 2026 The authors

This work is licensed under a Creative Commons Attribution 4.0 International License.









