TP53 Loss Fuels mTORC1 Activation and Autophagy Suppression to Drive Immune-Cold Colorectal Cancer
DOI:
https://doi.org/10.14740/wjon2695Keywords:
TP53, mTORC1, Autophagy, Colorectal cancer, Immunometabolism, Proteomics, Immune evasion, Regulatory T cellsAbstract
Background: Microsatellite-stable colorectal cancer (MSS CRC) is typically resistant to immune checkpoint blockade and remains an “immune-cold” disease. Wild-type p53 is known to restrain mTOR signaling and support autophagy, yet how TP53 loss integrates metabolic rewiring with immune suppression in MSS CRC—especially with protein- and phosphosite-level validation—remains incompletely defined. We tested whether p53 deficiency is associated with coordinated mTORC1 activation, autophagy attenuation, and immune-cold remodeling across multi-omics datasets.
Methods: We analyzed GSE146009 (paired tumor-normal RNA-seq pairs), TCGA-COAD/READ (n = 647, mutation annotated), GSE108989 (11,138 tumor-infiltrating T cells), and CPTAC colon proteome/phosphoproteome cohorts. Pathway activities were quantified by single-sample gene set enrichment analysis (ssGSEA) and Seurat module scoring. Group differences were tested by Kruskal–Wallis and Wilcoxon analyses with false-discovery-rate correction. CPTAC phosphosite-to-protein ratios were compared between TP53-mutant or p53-low versus wild-type or p53-high tumors.
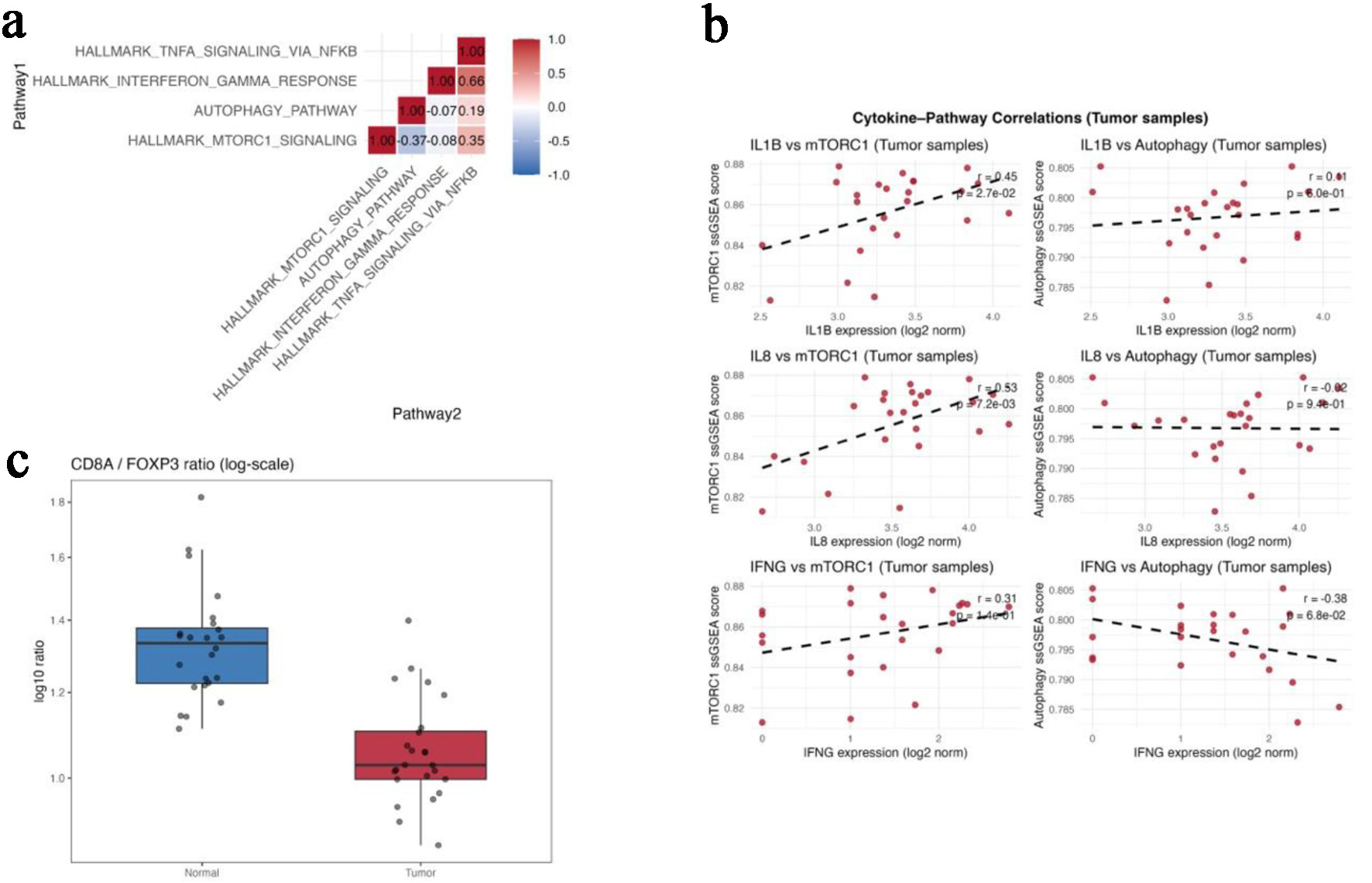
Results: Across bulk transcriptomes, TP53-mutant tumors showed higher mTORC1 signaling, lower autophagy scores, higher FOXP3, and reduced CD8A/FOXP3 ratios consistent with a regulatory T-cell-skewed immune-cold state. Single-cell analysis confirmed subset-specific immunometabolic programs, highlighting metabolically active regulatory/exhausted states with relatively blunted inflammatory output. Proteomic/phosphoproteomic data supported post-translational mTOR pathway activation in p53-deficient tumors, including increased phosphorylation of canonical mTOR substrates (EIF4EBP2 S65; RPS6KB1 T421/S424) and reduced phosphorylation at inhibitory RPTOR sites (S705/T725/S726), consistent with sustained mTORC1 activity.
Conclusions: In MSS CRC, p53 deficiency is consistently associated with immunometabolic remodeling marked by persistent mTOR pathway activation, relative autophagy attenuation, and FOXP3-dominant immune-cold features across transcriptomic, single-cell, and proteomic layers. These findings add protein- and phosphosite-level evidence linking TP53 loss to an immune-suppressive metabolic state and support biomarker-guided evaluation of mTOR kinase (TORC1/2) inhibition, which more fully suppresses 4E-BP/S6K phosphorylation, combined with PD-1/PD-L1 (± CTLA-4) blockade in TP53-deficient MSS CRC.
Published
Issue
Section
License
Copyright (c) 2026 The authors

This work is licensed under a Creative Commons Attribution 4.0 International License.









