| World Journal of Oncology, ISSN 1920-4531 print, 1920-454X online, Open Access |
| Article copyright, the authors; Journal compilation copyright, World J Oncol and Elmer Press Inc |
| Journal website https://wjon.elmerpub.com |
Original Article
Volume 16, Number 2, April 2025, pages 200-209
Patterns, Predictors, and Prognostic Implication of Treatment-Related Amenorrhea in Patients With Breast Cancer
Bryant Nga , Armeyra Devani
Ferintasaria
, Susanna Hilda Hutajulub,
i
, Yufi
Kartika Astaric
, Juan Adrian
Wiranatad
, Suwardjo Suwardjoe, Irianiwati
Widodof
, Lina Choridahg
, Wigati
Dhamiyatih
, Mardiah Suci
Hardiantib
, Kartika Widayati
Taroeno-Hariadib
aMedicine Study Program, Faculty of Medicine, Public Health and Nursing, Universitas
Gadjah Mada, Yogyakarta, Indonesia
bDivision of Hematology and Medical Oncology,
Department of Internal Medicine, Faculty of Medicine, Public Health and Nursing, Universitas
Gadjah Mada/Dr. Sardjito General Hospital Yogyakarta, Yogyakarta,
Indonesia
cDivision of Hematology and Medical Oncology, Department of Internal
Medicine, Dr. Sardjito General Hospital Yogyakarta, Yogyakarta,
Indonesia
dAcademic Hospital, Universitas Gadjah Mada, Yogyakarta,
Indonesia
eDivision of Surgical Oncology, Department of Surgery, Faculty of
Medicine, Public Health, and Nursing, Universitas Gadjah Mada/Dr. Sardjito General Hospital
Yogyakarta, Yogyakarta, Indonesia
fDepartment of Anatomical Pathology, Faculty
of Medicine, Public Health and Nursing, Universitas Gadjah Mada/Dr. Sardjito General Hospital
Yogyakarta, Yogyakarta, Indonesia
gDivision of Radiodiagnosis, Department of
Radiology, Faculty of Medicine, Public Health and Nursing, Universitas Gadjah Mada/Dr. Sardjito
General Hospital Yogyakarta, Yogyakarta, Indonesia
hDivision of Radiotherapy,
Department of Radiology, Faculty of Medicine, Public Health and Nursing, Universitas Gadjah
Mada/Dr. Sardjito General Hospital Yogyakarta, Yogyakarta,
Indonesia
iCorresponding Author: Susanna Hilda Hutajulu, Division of Hematology
and Medical Oncology, Department of Internal Medicine, Faculty of Medicine, Public Health and
Nursing, Universitas Gadjah Mada/Dr. Sardjito General Hospital, Yogyakarta 55284, Indonesia
Manuscript submitted November 9, 2024, accepted January 24, 2025, published online February 18,
2025
Short title: TRA in Breast Cancer Patients
doi: https://doi.org/10.14740/wjon1991
| Abstract | ▴Top |
Background: Treatment-related amenorrhea (TRA) is a common side effect of treatment in premenopausal patients with breast cancer, with important consequences for patient counseling and management. Its occurrence and potential influence on survival outcomes remain active areas of investigation. This study aimed to evaluate the incidence, risk factors, and prognostic significance of TRA in patients with breast cancer.
Methods: This is a retrospective cohort study. Patients were interviewed during and after chemotherapy to assess their menstrual status. Sociodemographic, clinical, and treatment data of patients were also collected. TRA was classified into early amenorrhea (EA) and late amenorrhea (LA) based on the duration of amenorrhea. Univariable and multivariable logistic regression were used to identify risk factors of EA and LA. Kaplan-Meier curves and Cox proportional hazards analyses were used to investigate the impact of EA and LA on 3-year overall survival (OS).
Results: There were 81 patients who were eligible for the final analysis. Of these subjects, 14 (17.3%) developed no amenorrhea, 67 (82.7%) developed EA, and 45 (55.6%) developed LA. We did not find any significant independent risk factor for EA. Age > 45 years (odds ratio (OR): 4.00; confidence interval (CI): 1.23 - 13.01; P = 0.021) and the usage of hormonal therapy (OR: 4.96; CI: 1.58 - 15.53; P = 0.006) independently significantly increase the risk of LA, whereas a metastatic disease status decreased the risk (OR: 0.20; CI: 0.04 - 0.90; P = 0.036). Both EA (hazard ratio (HR) = 0.262, CI: 0.105 - 0.653; P = 0.002) and LA (HR = 0.234, CI: 0.091 - 0.604; P = 0.001) were associated with an improved 3-year OS rate.
Conclusions: Age > 45 years and the usage of hormonal therapy are risk factors for LA, while metastatic disease was associated with a decreased risk. Both EA and LA had a significant association with favorable 3-year OS. These findings enable clinicians to provide personalized guidance, tailor treatment strategies, and improve the outcomes of premenopausal patients with breast cancer. Standardization of how TRA is defined and assessed in future studies is essential to improve comparability and enhance the understanding of its clinical implications.
Keywords: Breast cancer; Treatment-related amenorrhea; Risk factors; Survival
| Introduction | ▴Top |
Breast cancer (BC) remains the most frequently diagnosed cancer and the leading cause of cancer-related death among women worldwide, with an estimated age-standardized incidence rate of 46.8 per 100,000 and an estimated mortality rate of 12.7 per 100,000 [1]. Incidence rates demonstrate considerable variation, being higher in high-income countries but showing increasing trends in low- and middle-income countries [1, 2]. In Indonesia, BC is the most prevalent cancer among women, with an estimated age-standardized incidence rate of 41.8 per 100,000 and an estimated mortality rate of 14.4 per 100,000 [1]. Globally, survival outcomes of patients with BC differ by region, with more highly developed countries tending to have a higher survival rate. The 3-year survival rate and 5-year survival rate of patients with BC are estimated to be 75% and 73%, respectively [3]. However, survival data in Indonesia are limited, with an estimated 5-year survival rate ranging between 48% and 50% [4, 5].
BC treatment typically necessitates a multimodal approach, incorporating modalities such as chemotherapy, radiotherapy, and hormonal therapy, each of which is crucial in improving patient outcomes. However, these treatments are not without adverse effects, some of which may profoundly affect patients’ quality of life [6-8]. One such adverse effect is treatment-related amenorrhea (TRA). In previous studies, the incidence of TRA has been estimated to range between 15.1% and 98.3% [9], although heterogeneity in the definition of TRA may make interpretation challenging. This loss of menstrual function is not merely a marker of ovarian dysfunction but also has far-reaching physical and psychological consequences, including infertility, vasomotor symptoms [10], and a decreased quality of life [10, 11]. Managing TRA is especially crucial in Asian countries, given the higher prevalence of BC among premenopausal women [12]. This issue is also a significant concern particularly for patients with early-stage BC due to the typically favorable long-term survival outcomes [3].
The current landscape of research on TRA faces significant challenges due to inconsistencies in the definition of TRA, differences in symptom recording methods, and the variability of follow-up periods [13]. These issues do not only hinder our understanding of the true incidence, risk factors, and impact of TRA; they also complicate efforts to establish standardized guidelines for the diagnosis and management of patients with TRA.
The focus of this study is early amenorrhea (EA), as it may be more likely to be a result of treatment [14]. Another key focus is late amenorrhea (LA), as it can impose lasting impacts on a patient’s quality of life, fertility, and overall prognosis [11].
There remains a need for research on the risk factors [13] and prognostic significance of TRA [15], particularly in Indonesia, where such data are lacking. Therefore, our study aims to investigate the incidence, risk factors, and survival outcomes of TRA, with the goal of enhancing personalized treatment strategies, improving patient education, and contributing significantly to the existing body of literature.
| Materials and Methods | ▴Top |
Study design and subjects
This is a retrospective cohort study which included BC patients registered for the investigation of chemotherapy-related adverse effects and their impact on prognosis (main study). The inclusion criteria of the present study consisted of premenopausal women aged 18 - 55 with histopathologically confirmed stage I-IV BC and an Eastern Cooperative Oncology Group (ECOG) performance status of 0 - 1, who were scheduled for (neo)adjuvant first-line chemotherapy at the “Tulip”/Integrated Cancer Clinic, Dr. Sardjito General Hospital, Yogyakarta, Indonesia by the Hematology and Medical Oncology Division. Patients with terminal illnesses, severe comorbidities, or incomplete follow-up data were excluded. The study received ethical approval from the Joint Ethics Committee of the Faculty of Medicine, Public Health, and Nursing at Universitas Gadjah Mada/Dr. Sardjito General Hospital (KE/FK/0417/EC/2018). This study was conducted in compliance with the ethical standards of the responsible institution on human subjects, as well as with the Helsinki Declaration.
Study variables
Sociodemographic and clinical data were collected at the baseline upon patients’ inclusion in the main study. Sociodemographic data included in this study were age and parity. Subjects were grouped into ages ≤ 45 and ages > 45. The parity of the subjects, which was defined as the number of times a subject has given birth at a gestational age ≥ 24 weeks regardless of the condition of the fetus, was categorized into less than or equal to one (not multiparous) and more than one (multiparous). Clinical data used in this study were stage, estrogen receptor (ER) status, progesterone receptor (PR) status, and body mass index (BMI). Stage was defined as the stage of the patient at diagnosis according to the seventh edition of the American Joint Committee on Cancer (AJCC) staging system and was divided into stage I-III (not metastatic) and stage IV (metastatic), and ER and PR status were grouped as negative or positive. BMI was categorized as obese (≥ 25 kg/m2), overweight (23 - 24.9 kg/m2), normal (18.5 - 22.9 kg/m2), and underweight (< 18.5 kg/m2) in accordance with the World Health Organization (WHO) Asia-Pacific BMI classification system. Treatment data included in this study were radiotherapy, number of chemotherapy cycles, hormonal therapy, and chemotherapy regimen. Radiotherapy and hormonal therapy were grouped according to whether the subject received each or not. For the number of chemotherapy cycles, subjects were divided into whether they received less than eight chemotherapy cycles or eight or more chemotherapy cycles. Finally, the chemotherapy regimen of each subject was categorized to those who received an anthracycline-taxane regimen and those who did not.
Overall survival (OS) was calculated as the time in months between the initial diagnosis and death from any cause or the last follow-up. In this study, we used OS data assessed at a 3-year follow-up (3-year OS). We followed up patients’ status every 6 months through medical records and by phone.
Assessment of TRA
The status of amenorrhea was assessed using the Common Toxicity Criteria for Adverse Events (CTCAE) version 4.0 [16], which was translated into Bahasa Indonesia by practicing general practitioners and then reviewed by specialist physicians from the primary study [17]. A trained research team interviewed subjects 1 week after each cycle of chemotherapy and 1 week before the subsequent cycle to inquire about the occurrence and length of chemotherapy toxicities.
We classified the subjects’ posttreatment menstruation status of subjects into no amenorrhea, EA, and LA. Subjects with no amenorrhea had normal menses or resumption of menses within 6 months, whereas subjects with EA had an absence of menses for 6 months or more, and subjects with LA had an absence of menses for 12 months or more.
Statistical analysis
The number and percentage of subjects in the overall cohort by each of the sociodemographic, clinical, and treatment variables were calculated. The overall cohort is then stratified according to their posttreatment menstruation status (no amenorrhea, EA, and LA), and the number and percentage by each variable were also calculated. The median age of the subjects, as well as the mean and standard deviation of the BMI of the subjects were calculated for the overall cohort and the stratified cohorts.
To investigate risk factors for treatment-related EA and LA, we used logistic regression analyses. We applied univariable logistic regression by each sociodemographic, clinical, and treatment variables as independent variables, and LA and EA (EA versus no EA and LA versus no LA) as the dependent variable. The variance inflation factor (VIF) of each variable is calculated to analyze multicollinearity. Of these variables, those with a VIF of more than 2.5 were excluded from the multivariable logistic regression analysis, as this implies potential collinearity [18]. A P value of less than 0.05 was considered significant.
We conducted survival analysis to determine the effect of treatment-related EA and LA on the 3-year OS of the subjects. First, we plotted the Kaplan-Meier curve for the overall cohort, which illustrates the OS probability for the entire cohort from the time at diagnosis until the last follow-up. We calculated the shortest, median, and longest follow-up in the overall cohort. We then plotted Kaplan-Meier curves stratified according to whether the subjects had EA or not and then whether they had LA or not. We calculated the hazard ratio (HR) using an univariable Cox proportional hazards model and determined the P value using the log-rank test.
All statistical analyses were performed using R statistical software (v4.4.1; R Core Team 2024) [19]. Survival analysis was conducted using the R packages “survival” [20] and “survminer” [21].
| Results | ▴Top |
Subject characteristics
There were 249 subjects who were recruited in the main study. Of these, 144 premenopausal subjects aged ≤ 55 were included. A total of 35 subjects who did not receive chemotherapy and 11 subjects who received less than three cycles of chemotherapy were excluded. Moreover, 10 subjects were excluded due to not having menstruation status follow-up after chemotherapy. Finally, seven subjects who were followed for less than 3 years were excluded, leaving 81 eligible subjects to be included in the final analysis of the present study (Supplementary Material 1, wjon.elmerpub.com). The majority of the subjects were aged 45 or below (55.6%), were multiparous (70.4%), had non-metastatic disease at diagnosis (80.2%), had ER-positive disease (67.9%), had PR-positive (56.8%) disease, had normal BMI (39.5%), received radiotherapy (72.8%), underwent eight or more chemotherapy cycles (63.0%), did not receive an anthracycline-taxane regimen (88.9%), and received hormonal therapy (64.2%) (Table 1).
 Click to view |
Table 1. Subject Demographics and Clinical
Characteristics |
Patterns of TRA
Of the 81 subjects, 14 (17.3%) developed no amenorrhea, 67 (82.7%) developed EA, and 45 (55.6%) developed LA. Of the 67 subjects with EA, 45 (67.2%) progressed to LA (Table 1).
The median age of all subjects was 45.4 years. Among the 45 subjects aged ≤ 45 years, 10 (22.2%) developed no amenorrhea, 35 (77.8%) developed EA, and 18 (40.0%) developed LA. Among the 36 subjects aged > 45 years, four developed no amenorrhea (11.1%), 32 (88.9%) developed EA, and 27 (75.0%) developed LA (Table 1).
Predictors of TRA
Univariable logistic regression showed five independent variables with a P value < 0.250, which served as potential predictors of EA. These variables are age > 45 years (odds ratio (OR): 2.29; confidence interval (CI): 0.65 - 8.02; P = 0.197), stage IV disease (OR: 0.35; CI: 0.10 - 1.26; P = 0.109), receiving radiotherapy (OR: 3.19; CI: 0.97 - 10.46; P = 0.056), receiving eight or more chemotherapy cycles (OR: 3.94; CI: 1.18 - 13.21, P = 0.026), and receiving hormonal therapy (OR: 2.92; CI: 0.90 - 9.48; P = 0.074) (Table 2). The stage, ER and PR status of subjects were excluded from the multivariable analysis as they had a VIF of more than 2.5 (Supplementary Material 2, wjon.elmerpub.com). However, none of the variables included in the multivariable regression proved to be significant predictors of EA (Table 2).
 Click to view |
Table 2. Sociodemographic and Clinical Factors
Associated With Early Amenorrhea After Breast Cancer Treatment |
In the univariable logistic regression analysis of LA, seven independent variables had a P value < 0.250, and hence are potential predictors of LA. These variables are age > 45 years (OR: 4.50; CI: 1.72 - 11.77; P = 0.002), parity > 1 (OR: 0.40; CI: 0.14 - 1.10; P = 0.077), stage IV disease (OR: 0.13; CI: 0.03 - 0.49; P = 0.003), ER-positive status (OR: 3.58; CI: 1.34 - 9.54; P = 0.011), PR-positive status (OR: 2.50, CI: 1.01 - 6.17; P = 0.047), receiving eight or more chemotherapy cycles (OR: 2.75; CI: 1.09 - 6.96, P = 0.033), and receiving hormonal therapy (OR: 5.00; CI: 1.87 - 13.36; P = 0.001) (Table 3). The ER and PR status were excluded from the multivariable analysis as they had a VIF of more than 2.5 (Supplementary Material 3, wjon.elmerpub.com).
 Click to view |
Table 3. Sociodemographic and Clinical Factors
Associated With Late Amenorrhea After Breast Cancer Treatment |
The multivariable logistic regression showed that age > 45 (OR: 4.00; CI: 1.23 - 13.01; P = 0.021) and the usage of hormonal therapy (OR: 4.96; CI: 1.58 - 15.53; P = 0.006) increased the risk of LA. On the other hand, a metastatic disease status was protective against LA (OR: 0.20; CI: 0.04 - 0.90; P = 0.036) (Table 3).
Prognostic significance of TRA
The median follow-up duration was 54.4 months (range: 6.3 - 75.0 months). The Kaplan-Meier survival curve for the overall cohort showed a decrease in survival probability throughout the follow-up period. The survival probability was 97.5% at 12 months, 73.0% at 36 months, 69.8% at 60 months, and 66.9% at 72 months (Fig. 1a).
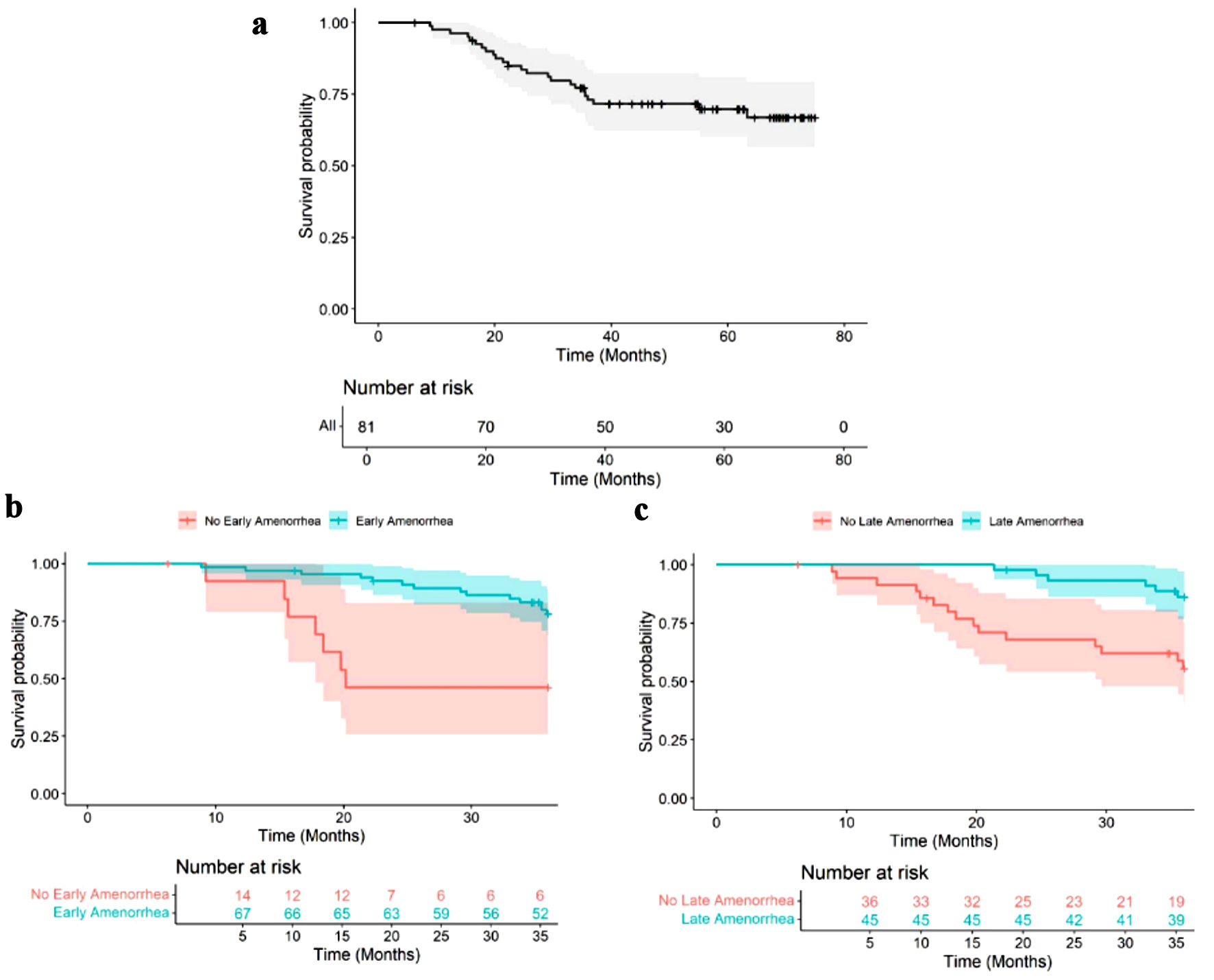 Click for large image |
Figure 1. Kaplan-Meier survival curves for the overall cohort and for comparing patients according to their TRA status. (a) Kaplan-Meier curve for the overall cohort. (b) Kaplan-Meier curves comparing survival of patients with EA to patients without EA. (c) Kaplan-Meier survival curves comparing survival of patients with LA to patients without LA. TRA: treatment-related amenorrhea; EA: early amenorrhea; LA: late amenorrhea. |
The Kaplan-Meier survival curves comparing patients with EA against patients without EA showed a significant divergence in OS. The EA group had a higher 2-year (92.5% vs. 46.2%) and 3-year (78.2% vs. 46.2%) survival rate. Cox regression analysis revealed that EA was associated with a significantly lower risk of death (HR = 0.262, CI: 0.105 - 0.653; P = 0.002), conveying a protective effect of EA on survival (Fig. 1b).
The Kaplan-Meier survival curves also showed that the LA group had a higher 2-year (97.8% vs. 68.0%) and 3-year (86.3% vs. 55.5%) survival rate compared to patients without LA. Cox regression analysis showed that LA was also associated with a significantly lower risk of death (HR = 0.234, CI: 0.091 - 0.604; P = 0.001), implying that LA also has protective effects on survival (Fig. 1c).
| Discussion | ▴Top |
Study novelty and primary findings
TRA in Indonesian patients with BC is rarely investigated. Given the limited data on this topic, this study fills an important gap in understanding the incidence, risk factors, and prognostic significance of TRA. Through the examination of both EA and LA, this study provides a comprehensive view on the occurrences of different durations of TRA, as well as their impact on survival.
Comparison to previous literature
There is a lack of consensus in describing the phenomenon of amenorrhea which occurs after starting the treatment of BC. Indeed, different terms, such as chemotherapy-induced amenorrhea [22-28], chemotherapy-related amenorrhea [11, 29, 30], and TRA [15, 31, 32] have been used. We opted to use TRA, as modalities other than chemotherapy, such as hormonal therapy [7, 11, 27, 31] and radiotherapy [33], may also cause amenorrhea. Various durations of lack of menses have also been used as cutoffs, including 3 months [22, 27-29], 6 months [15, 23, 24, 30, 31, 34], 12 months [25-29], and 18 months [31]. Therefore, we investigated using both 6 or more months (EA) and 12 or more months (LA) as cutoffs in our study.
The incidence of EA in our study (82.7%) appears to be similar to that reported in a study in the USA (79.7%) [34], slightly higher than that in a study in Belgium (72.1-74.8%) [15] and in Japan (72.7%) [24]. The incidence rate in our report is considerably higher than in other studies in the USA, with one study reporting incidence rates of 24-50% [30], and another reporting an incidence of 36.2% [31]. The incidence of LA in our study (55.6%) is similar to what was reported in a study in Korea (57.7%) [27] and in Iran (58.8%) [25], considerably lower than a study in Pakistan (91.5%) [26], and slightly higher than another study in Indonesia (45.7%) [28] and one study in China (48.9%) [29]. The differences may reflect variations in genetic factors, healthcare settings, or treatment approaches warranting further investigation to determine their specific roles in influencing the incidence of TRA. Inconsistent definitions and follow-up periods across studies complicate direct comparisons, emphasizing the need for standardized methodologies in future research on TRA.
We found that age > 45 is a predictor of LA. This is consistent with previous studies, which report that the higher the age, the higher the risk of TRA [11, 24, 27-29, 32]. This effect may be due to the physiological decline in the number and quality of oocytes in the ovaries with aging [35]. Additionally, this may reflect an increased ovarian sensitivity to the cytotoxic effects of chemotherapy in older patients [9].
The usage of hormonal therapy increased the risk of LA, which is similar to what previous studies reported [11, 27, 31]. Hormonal therapies may disrupt ovarian function by reducing estrogen levels or blocking ERs [36], which may induce or prolong amenorrhea.
On the other hand, a metastatic disease status was protective against LA. An explanation for this finding is that patients with metastatic BC tended to receive shorter treatment courses and hence their ovaries had a lower cumulative exposure to cytotoxic agents, reducing the probability of amenorrhea (Supplementary Material 4, wjon.elmerpub.com). Further investigation is needed to explore potential biological and treatment-related mechanisms behind this protective effect.
Both EA and LA had a protective prognostic effect on the 3-year OS of patients, with LA having a slightly stronger effect. This finding is consistent with previous studies [15, 23, 34]. This suggests that the occurrence of TRA may reflect a stronger therapeutic response to treatment and indicate more effective ovarian suppression, contributing to more favorable survival outcomes [9]. Additionally, this protective effect may also be linked to a decrease in circulating estrogen levels, which may inhibit the growth and progression of BC [37].
Clinical implications
This study has important clinical implications, particularly in the management of BC in premenopausal women. Our finding that TRA may be associated with better survival suggests that TRA may be used as a prognostic marker to stratify patients based on their likelihood of survival. However, TRA may lead to negative effects such as infertility and a diminished quality of life [11]. We identified key risk factors for the occurrence of TRA. This enables clinicians to better assess the risk of TRA in their patients, develop personalized treatment plans, and anticipate the need for symptom management. Incorporating discussions of the likelihood of TRA may guide patient expectations, improve treatment adherence, and address potential symptom management needs.
Study strengths and limitations
A strength of this study is the prospective gathering of data from patients with regular follow-up intervals, which enabled longitudinal monitoring of menstrual status. Furthermore, the usage of a trained interviewer in favor of a questionnaire enabled two-way communication for clarification of questions or responses, which ensured accurate gathering of data and reduced misreporting. These allowed for a more reliable assessment of TRA over time, further strengthening the validity of our study’s findings.
This study has several limitations. We did not utilize a menopause-specific scoring system or perform an endocrine profile assay, which could have offered a more accurate assessment of the subjects’ menopausal status. Moreover, this analysis did not consider some factors, such as cumulative dose, smoking, alcohol consumption, exercise, as well as stress levels and psychological factors, which may influence the occurrence of TRA and are potential subjects for future investigations. Lastly, the study’s single-center design may limit the generalizability of these findings.
Conclusions
An age > 45 and the usage of hormonal therapy were independent predictors of LA, while metastatic disease was associated with a decreased risk. Both EA and LA were associated with improved 3-year OS. These findings underscore the potential role of TRA as a prognostic marker and emphasize the need for clinicians to consider TRA in personalized treatment planning and patient counseling. Further research is needed to better understand the mechanisms underlying these findings and to optimize management of patients with BC patients who are at risk of TRA. Our study also highlights the need for standardization of the definition and assessment of TRA to improve consistency across future research. Such standardization would allow for better comparisons of results and more robust conclusions.
| Supplementary Material | ▴Top |
Suppl 1. Flowchart of subject selection for final analysis.
Suppl 2. Variance inflation factors of the independent variables of the logistic regression analysis with early amenorrhea as the dependent variable.
Suppl 3. Variance inflation factors of the independent variables of the logistic regression analysis with late amenorrhea as the dependent variable.
Suppl 4. Number of chemotherapy cycles according to metastatic disease status.
Acknowledgments
We thank Benedreky Leo, Irfan Haris, Riani Witaningrum, Norma Dewi Suryani, and Betrix Rifana for technical assistance.
Financial Disclosure
This study was funded by Kementrian Riset Teknologi dan Pendidikan Tinggi Republik Indonesia (ID) (2020, grant number: 2258/UN1/DITLIT/DIT-LIT/PT/2020) and Universitas Gadjah Mada (2023, grant number: 1642/UN1/FKKMK.1.3/PPKE/PT/2023).
Conflict of Interest
The author(s) declare that they have no conflict of interest.
Informed Consent
Written informed consent was obtained from all study participants.
Author Contributions
Conception and design: Bryant Ng, Susanna Hilda Hutajulu, Juan Adrian Wiranata. Administrative support: Susanna Hilda Hutajulu, Yufi Kartika Astari, Juan Adrian Wiranata. Provision of study materials or patients: Susanna Hilda Hutajulu, Suwardo Suwardjo, Irianiwati Widodo, Lina Choridah, Wigati Dhamiyati, Mardiah Suci Hardianti, Kartika Widayati Taroeno-Hariadi. Collection and assembly of data: Bryant Ng, Armeyra Devani Ferintasari, Susanna Hilda Hutajulu, Yufi Kartika Astari. Data analysis and interpretation: all authors. Manuscript writing: all authors. Final approval of manuscript: all authors.
Data Availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
| References | ▴Top |
- Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I,
Jemal A, Bray F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality
worldwide for 36 cancers in 185 countries. CA Cancer J Clin.
2021;71(3):209-249.
doi pubmed - Lv L, Zhao B, Kang J, Li S, Wu H. Trend of disease burden
and risk factors of breast cancer in developing countries and territories, from 1990 to 2019:
Results from the Global Burden of Disease Study 2019. Front Public Health.
2022;10:1078191.
doi pubmed - Maajani K, Jalali A, Alipour S, Khodadost M, Tohidinik HR,
Yazdani K. The global and regional survival rate of women with breast cancer: a systematic
review and meta-analysis. Clin Breast Cancer. 2019;19(3):165-177.
doi pubmed - Sinaga ES, Ahmad RA, Shivalli S, Hutajulu SH. Age at
diagnosis predicted survival outcome of female patients with breast cancer at a tertiary
hospital in Yogyakarta, Indonesia. Pan Afr Med J. 2018;31:163.
doi pubmed - Terhell AJ, Wahyuni S, Pryce A, Koot JW, Abadi K,
Yazdanbakhsh M. Anti-filarial and total IgG4 and IgE antibody levels are correlated in mothers
and their offspring. Trans R Soc Trop Med Hyg. 2002;96(3):334-339.
doi pubmed - Behranvand N, Nasri F, Zolfaghari Emameh R, Khani P,
Hosseini A, Garssen J, Falak R. Chemotherapy: a double-edged sword in cancer treatment. Cancer
Immunol Immunother. 2022;71(3):507-526.
doi pubmed - Berkowitz MJ, Thompson CK, Zibecchi LT, Lee MK, Streja E,
Berkowitz JS, Wenziger CM, et al. How patients experience endocrine therapy for breast cancer:
an online survey of side effects, adherence, and medical team support. J Cancer Surviv.
2021;15(1):29-39.
doi pubmed - Dilalla V, Chaput G, Williams T, Sultanem K. Radiotherapy
side effects: integrating a survivorship clinical lens to better serve patients. Curr Oncol.
2020;27(2):107-112.
doi pubmed - Wang Y, Li Y, Liang J, Zhang N, Yang Q. Chemotherapy-induced
amenorrhea and its prognostic significance in premenopausal women with breast cancer: an updated
meta-analysis. Front Oncol. 2022;12:859974.
doi pubmed - Schover LR. Premature ovarian failure and its consequences:
vasomotor symptoms, sexuality, and fertility. J Clin Oncol. 2008;26(5):753-758.
doi pubmed - Kabirian R, Franzoi MA, Havas J, Coutant C, Tredan O, Levy
C, Cottu P, et al. Chemotherapy-related amenorrhea and quality of life among premenopausal women
with breast cancer. JAMA Netw Open. 2023;6(11):e2343910.
doi pubmed - Cheng SH, Tsou MH, Liu MC, Jian JJ, Cheng JC, Leu SY, Hsieh
CY, et al. Unique features of breast cancer in Taiwan. Breast Cancer Res Treat.
2000;63(3):213-223.
doi pubmed - Zavos A, Valachis A. Risk of chemotherapy-induced amenorrhea
in patients with breast cancer: a systematic review and meta-analysis. Acta Oncol.
2016;55(6):664-670.
doi pubmed - Goodwin PJ, Ennis M, Pritchard KI, Trudeau M, Hood N. Risk
of menopause during the first year after breast cancer diagnosis. J Clin Oncol.
1999;17(8):2365-2370.
doi pubmed - Lambertini M, Campbell C, Bines J, Korde LA, Izquierdo M,
Fumagalli D, Del Mastro L, et al. Adjuvant anti-HER2 therapy, treatment-related amenorrhea, and
survival in premenopausal HER2-positive early breast cancer patients. J Natl Cancer Inst.
2019;111(1):86-94.
doi pubmed - Common Terminology Criteria for Adverse Events (CTCAE) Version 4.0. U.S. Department of Health and Human Services. (2010). Accessed: October 02, 2024: https://evs.nci.nih.gov/ftp1/CTCAE/CTCAE_4.03/Archive/CTCAE_4.0_2009-05-29_QuickReference_8.5x11.pdf.
- Wiranata JA, Hutajulu SH, Astari YK, Leo B, Bintoro BS,
Hardianti MS, Taroeno-Hariadi KW, et al. Patient-reported outcomes and symptom clusters pattern
of chemotherapy-induced toxicity in patients with early breast cancer. PLoS One.
2024;19(2):e0298928.
doi pubmed - Johnston R, Jones K, Manley D. Confounding and collinearity
in regression analysis: a cautionary tale and an alternative procedure, illustrated by studies
of British voting behaviour. Qual Quant. 2018;52(4):1957-1976.
doi pubmed - R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria. 2024. https://www.R-project.org/.
- Terry M, Therneau PM. Modeling survival data: extending cox model. New York: Springer; 2000.
- Kassambara A, Kosinski M, Biecek P. Survminer: Drawing Survival Curves using 'ggplot2'. R package version 0.4.9. 2021. https://CRAN.R-project.org/package=survminer.
- Eivazi-Ziaei J, Asvadi I, Esfahani A, Vaez J, Pourzand A,
Shabanloei R, Sattari MR. Chemotherapy-induced amenorrhea in patients suffering from breast
cancer in the Northwest of Iran. South Asian J Cancer. 2014;3(4):233-234.
doi pubmed - Ferretti G, Carlini P, Bria E, Felici A, Giannarelli D,
Ciccarese M, Papaldo P, et al. Chemotherapy-induced amenorrhea in early breast cancer. Ann
Oncol. 2006;17(2):352.
doi pubmed - Iwamoto T, Hara F, Uemura Y, Mukai H, Watanabe T, Ohashi Y.
NSAS-BC02 substudy of chemotherapy-induced amenorrhea (CIA) in premenopausal patients who
received either taxane alone or doxorubicin(A) cyclophosphamide(C) followed by taxane as
postoperative chemotherapy. Breast Cancer Res Treat. 2020;182(2):325-332.
doi pubmed - Pourali L, Taghizadeh Kermani A, Ghavamnasiri MR, Khoshroo
F, Hosseini S, Asadi M, Anvari K. Incidence of chemotherapy-induced amenorrhea after adjuvant
chemotherapy with taxane and anthracyclines in young patients with breast cancer.
Iran J Cancer Prev. 2013;6(3):147-150.
pubmed - Ravi R, Haider G, Ahmed K, Sami A, Zahoor S, Lata R.
Amenorrhea After Chemotherapy In Breast Cancer Patient. J Ayub Med Coll Abbottabad.
2020;32(1):73-77.
pubmed - Yoo C, Yun MR, Ahn JH, Jung KH, Kim HJ, Kim JE, Park JY, et
al. Chemotherapy-induced amenorrhea, menopause-specific quality of life, and endocrine profiles
in premenopausal women with breast cancer who received adjuvant anthracycline-based
chemotherapy: a prospective cohort study. Cancer Chemother Pharmacol.
2013;72(3):565-575.
doi pubmed - Mudib M, Yarso KY, Nugroho HA. Age as an independent factor in the permanent Chemotherapy-Induced Amenorrhea incidence in breast cancer patients at Dr. Moewardi Hospital, Surakarta, Indonesia. Open Access Maced J Med Sci. 2021;9(B):816-820.
- Liem GS, Mo FK, Pang E, Suen JJ, Tang NL, Lee KM, Yip CH, et
al. Chemotherapy-related amenorrhea and menopause in young chinese breast cancer patients:
analysis on incidence, risk factors and serum hormone profiles. PLoS One.
2015;10(10):e0140842.
doi pubmed - Ruddy KJ, Zheng Y, Tayob N, Hu J, Dang CT, Yardley DA,
Isakoff SJ, et al. Chemotherapy-related amenorrhea (CRA) after adjuvant ado-trastuzumab
emtansine (T-DM1) compared to paclitaxel in combination with trastuzumab (TH) (TBCRC033: ATEMPT
Trial). Breast Cancer Res Treat. 2021;189(1):103-110.
doi pubmed - Poorvu PD, Hu J, Zheng Y, Gelber SI, Ruddy KJ, Tamimi RM,
Peppercorn JM, et al. Treatment-related amenorrhea in a modern, prospective cohort study of
young women with breast cancer. NPJ Breast Cancer. 2021;7(1):99.
doi pubmed - Shin JJ, Choi YM, Jun JK, Lee KH, Kim TY, Han W, Im SA.
Amenorrhea and menopause in patients with breast cancer after chemotherapy. J Breast
Cancer. 2019;22(4):624-634.
doi pubmed - Kim S, Kim SW, Han SJ, Lee S, Park HT, Song JY, Kim T.
Molecular mechanism and prevention strategy of chemotherapy- and radiotherapy-induced ovarian
damage. Int J Mol Sci. 2021;22(14):7484.
doi pubmed - Swain SM, Jeong JH, Geyer CE, Jr., Costantino JP, Pajon ER,
Fehrenbacher L, Atkins JN, et al. Longer therapy, iatrogenic amenorrhea, and survival in early
breast cancer. N Engl J Med. 2010;362(22):2053-2065.
doi pubmed - Park SU, Walsh L, Berkowitz KM. Mechanisms of ovarian aging.
Reproduction. 2021;162(2):R19-R33.
doi pubmed - Johnston SJ, Cheung KL. Endocrine therapy for breast cancer:
a model of hormonal manipulation. Oncol Ther. 2018;6(2):141-156.
doi pubmed - Al-Shami K, Awadi S, Khamees A, Alsheikh AM, Al-Sharif S,
Ala' Bereshy R, Al-Eitan SF, et al. Estrogens and the risk of breast cancer: A narrative review
of literature. Heliyon. 2023;9(9):e20224.
doi pubmed
This
article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0
International License, which permits unrestricted non-commercial use, distribution, and
reproduction in any medium, provided the original work is properly cited.
World
Journal of Oncology is published by Elmer Press Inc.