| World Journal of Oncology, ISSN 1920-4531 print, 1920-454X online, Open Access |
| Article copyright, the authors; Journal compilation copyright, World J Oncol and Elmer Press Inc |
| Journal website https://wjon.elmerpub.com |
Original Article
Volume 17, Number 3, June 2026, pages 419-427
Immunohistochemistry as a Cornerstone in Lung Cancer Diagnosis: Subtyping Based on the 2021 World Health Organization Classification and Its Practical Application
Imrana Tanvira, Amber Hassanb, l, Bushra Nisarc, Sadia Khand, Maryam Altafd, Humaira Waseeme, Hussain Noorwalif, Ali A. Mousag, Amany Fathaddinh, Ragdah Arifi, Mohammed M. Karamij, Majid Almansourik, l
aDepartment of Pathology, Faculty of Medicine, King Abdulaziz University, Rabigh, Saudi Arabia
bEuropean School of Molecular Medicine, University of Milan, Milan, Italy
cAmeerudeen Medical College/PGMI/LGH Lahore, Lahore, Pakistan
dBPP University, Manchester, UK
eFatima Jinnah Medical University, Lahore, Pakistan
fDepartment of Basic Medical Sciences, College of Medicine, University of Jeddah, Jeddah, Saudi Arabia
gDepartment of Anatomy and Embryology, Faculty of Medicine, Jouf University, Jouf, Saudi Arabia
hDepartment of Pathology, Faculty of Medicine, King Saud University, Riyadh, Saudi Arabia
iDepartment of Internal Medicine, Faculty of Medicine, King Abdulaziz University, Jeddah, Saudi Arabia
jDepartment of Clinical Physiology, Faculty of Medicine, King Abdulaziz University, Jeddah, Saudi Arabia
kDepartment of Clinical Biochemistry, Faculty of Medicine, King Abdulaziz University, Jeddah, Saudi Arabia
lCorresponding Authors: Majid Almansouri, Department of Clinical Biochemistry, Faculty of Medicine, King Abdulaziz University, Jeddah, Saudi Arabia; Amber Hassan, European School of Molecular Medicine, University of Milan, Milan, Italy
Manuscript submitted March 3, 2026, accepted April 15, 2026, published online May 8, 2026
Short title: IHC in Lung Tumor Subclassification
doi: https://doi.org/10.14740/wjon2757
| Abstract | ▴Top |
Background: Lung cancer is the leading cause of cancer-related deaths worldwide, and precise histological classification is vital for therapy decisions. Given diagnostic limitations with small biopsies, immunohistochemistry (IHC) enhances accuracy. This study evaluated the diagnostic value of IHC markers in subtyping lung tumors per the 2021 World Health Organization (WHO) classification and differentiating primary from metastatic lesions.
Methods: A prospective study was conducted on 151 lung biopsy specimens over 18 months. All samples were processed using standard histopathological techniques and an IHC panel comprising thyroid transcription factor-1 (TTF-1), napsin A, cytokeratin (CK)7, CK5/6, P63, synaptophysin, and chromogranin. Additional markers (estrogen receptor (ER), progesterone receptor (PR), human epidermal growth factor receptor 2 (HER2), PAX-8, PAX-2, CD10) were applied for metastatic differentiation. Statistical correlation analysis between lineage-specific markers was performed to assess diagnostic concordance.
Results: Adenocarcinoma was the most prevalent subtype (58.3%), followed by neuroendocrine neoplasms (17.2%), metastatic lesions (13.2%), adenosquamous carcinoma (6.0%), and squamous cell carcinoma (2.6%). Rare entities included carcinosarcoma and mucoepidermoid carcinoma (each 1.3%). TTF-1 and napsin A were highly specific for adenocarcinoma (85% and 94%, respectively), whereas CK5/6 and P63 confirmed squamous differentiation (100% each). Synaptophysin and chromogranin were positive in 96% and 100% of neuroendocrine tumors, respectively. Correlation analysis demonstrated strong marker concordance: TTF-1 vs napsin A (r = 0.88, P < 0.001), P63 vs CK5/6 (r = 0.93, P < 0.001), and synaptophysin vs chromogranin (r = 0.87, P < 0.001). All metastatic lesions were TTF-1 negative, confirming their extrapulmonary origin.
Conclusions: Combining IHC with histopathology markedly improves diagnostic precision and consistency in lung tumor classification. Lineage-specific markers validate the IHC panel’s reliability, distinguishing adenocarcinoma, squamous, and neuroendocrine types while differentiating primary from metastatic lesions. This integrative approach supports WHO-aligned precision diagnostics and guides individualized therapeutic strategies.
Keywords: Lung carcinoma; Immunohistochemistry; Adenocarcinoma; Squamous cell carcinoma; Neuroendocrine tumor; WHO classification; Metastasis
| Introduction | ▴Top |
Lung cancer remains the leading cause of cancer-related mortality worldwide, accounting for nearly one in five cancer deaths annually [1]. Histologically, lung tumors represent a heterogeneous group of malignancies arising from epithelial, mesenchymal, or neuroendocrine origins [2]. Among these, non-small cell lung carcinoma (NSCLC) constitutes approximately 85% of all lung cancers. It is further classified into adenocarcinoma, squamous cell carcinoma, and large cell carcinoma, each characterized by distinct morphological and molecular features [3–5].
In recent decades, adenocarcinoma has surpassed squamous cell carcinoma as the predominant histological subtype, a trend attributed to changing smoking habits, environmental exposures, and genetic susceptibility [6, 7]. The latest World Health Organization (WHO) classification of lung tumors integrates histopathological, molecular, and radiological criteria to enable accurate subclassification and to guide targeted therapeutic strategies [5, 8].
Despite advances in radiological imaging and minimally invasive diagnostic techniques, a large proportion of lung cancers are diagnosed at advanced stages when only limited biopsy or cytological material is available [9–11]. Under these circumstances, morphological evaluation alone is often insufficient, especially in poorly differentiated or overlapping histological patterns [4, 12]. Therefore, immunohistochemistry (IHC) has become an indispensable adjunct to conventional histopathology, providing lineage-specific information that refines diagnosis and informs management [13].
IHC enables accurate subtyping of NSCLC and facilitates differentiation from other tumor entities, including neuroendocrine and metastatic lesions [11, 14]. Markers such as thyroid transcription factor-1 (TTF-1) and napsin A are specific for adenocarcinoma, whereas cytokeratin (CK)5/6, P63, and its isoform P40 reliably indicate squamous differentiation [15–17]. Synaptophysin and chromogranin are essential markers of neuroendocrine differentiation, whereas CK7 expression is observed in most epithelial tumors and supports lineage determination [18].
While the diagnostic role of individual immunohistochemical markers is well established, their combined performance and concordance in routine clinical practice, particularly in small biopsy specimens, remain critically relevant. In real-world diagnostic settings, limited tissue availability often restricts morphological assessment, necessitating reliance on optimized immunohistochemical panels [10, 19]. This study therefore provides a systematic, data-driven evaluation of a focused IHC panel within the framework of the WHO 2021 classification, with emphasis on marker concordance, diagnostic reliability, and practical applicability in routine pathology workflows, including the distinction between primary and metastatic lesions [20–22].
| Materials and Methods | ▴Top |
Study design and duration
This prospective descriptive study was conducted in the Department of Pathology at a tertiary care teaching hospital over 18 months, from February 2023 to December 2024. A total of 151 lung biopsy specimens were included based on histopathological suspicion or radiological evidence of malignancy. Ethical approval for the study protocol was obtained from the Institutional Review Board, and the study was conducted in accordance with the Declaration of Helsinki.
Sample selection and criteria
A total of 151 consecutive lung biopsy specimens were included, comprising both bronchoscopic and transthoracic needle core biopsies. Only cases showing malignant pulmonary lesions with adequate tissue for histopathological and IHC evaluation were enrolled. Non-neoplastic, inflammatory, or inadequate biopsies were excluded from the study. Relevant clinical and radiological data were retrieved from patient records to aid in diagnostic correlation.
Tissue processing and histopathological examination
Biopsy samples were immediately fixed in 10% neutral buffered formalin for 6–24 h and processed through graded alcohols for dehydration, followed by xylene for clearing and paraffin embedding. Tissue blocks were sectioned at 3–5 µm thickness using a rotary microtome. Routine hematoxylin and eosin (H&E) staining was performed to assess tumor morphology and to establish a preliminary diagnosis.
Immunohistochemical analysis
Immunohistochemical staining was carried out on formalin-fixed, paraffin-embedded (FFPE) tissue sections using the standard streptavidin–biotin peroxidase technique. Sections were deparaffinized in xylene, rehydrated through descending grades of alcohol, and subjected to antigen retrieval in citrate buffer (pH 6.0) using a microwave-based protocol. Endogenous peroxidase activity was blocked using 3% hydrogen peroxide. The primary antibody panel included TTF-1 (clone 8G7G3/1) and napsin A as markers for adenocarcinoma differentiation; CK7 as an epithelial marker for NSCLC; CK5/6 and P63 as markers for squamous cell carcinoma differentiation; and synaptophysin and chromogranin as neuroendocrine differentiation markers.
In selected cases with suspected metastasis, additional site-specific markers were used, including estrogen receptor (ER), progesterone receptor (PR), human epidermal growth factor receptor 2 (HER2), PAX-8, CD10, and E-cadherin to identify primary sites such as breast, renal, or oral cavity carcinomas [14, 19]. Immunoreactivity was visualized using diaminobenzidine (DAB) as the chromogen and counterstained with Mayer’s hematoxylin. Positive and negative controls were included with each staining batch to ensure accuracy and reproducibility. The expression pattern for each marker was evaluated semi-quantitatively based on staining intensity and the percentage of tumor cells stained.
Diagnostic categorization
All cases were reviewed independently by two experienced histopathologists. Final diagnoses were established by integrating morphological features and immunohistochemical profiles in accordance with the 2021 WHO classification of thoracic tumors [5].
Statistical analysis
All data were analyzed using IBM SPSS Statistics version 26.0 (IBM Corp., Armonk, NY, USA). Descriptive statistics were applied to summarize demographic and pathological variables. Categorical data, including age group, gender distribution, histopathological subtype, and IHC expression patterns, were presented as frequencies and percentages.
The diagnostic specificity of each IHC marker was determined by correlating its staining profile with the final histopathological diagnosis. Pearson’s correlation coefficient (r) was calculated to assess the strength and direction of associations between lineage-specific markers, including TTF-1 vs napsin A, P63 vs CK5/6, and synaptophysin vs chromogranin. A P value < 0.05 was considered statistically significant, while correlations with P < 0.001 were interpreted as highly significant.
| Results | ▴Top |
Demographic profile
A total of 151 lung tumor cases were analyzed over 18 months. Patient ages ranged from 30 to 82 years (mean ± standard deviation (SD), 61.8 ± 9.4 years). The highest incidence was observed in the 51–60-year age group (36.4%), followed by the 61–70-year age group (33.1%) and the > 70-year age group (18.5%). Patients aged 41–50 years accounted for 9.3% of cases, while those aged 30–40 years accounted for 2.7%. The study population exhibited a clear male predominance, comprising 103 males (68.2%) and 48 females (31.8%), yielding a male-to-female ratio of 2.14:1
Primary versus metastatic lesions
Of the 151 cases, 131 (86.8%) were primary lung tumors, and 20 (13.2%) were secondary metastatic deposits. The most common sources of metastasis included the breast (10 cases), kidney (six cases), and oral cavity (four cases). All metastatic lesions were morphologically confirmed and further validated by IHC using organ-specific markers.
Histopathological spectrum
Adenocarcinoma was the predominant subtype, observed in 88 cases (58.3%), followed by neuroendocrine neoplasms (26 cases, 17.2%), and metastatic lesions (20 cases, 13.2%). Other less frequent subtypes included adenosquamous carcinoma (nine cases, 6.0%), squamous cell carcinoma (four cases, 2.6%), and rare entities such as carcinosarcoma and mucoepidermoid carcinoma (each 1.3%) (Table 1).
 Click to view | Table 1. Distribution of Lung Tumor Subtypes (N = 151) |
Histopathological examination of adenocarcinoma revealed malignant epithelial cells arranged in glandular and papillary configurations with occasional solid nests. Tumor cells exhibited pleomorphic, hyperchromatic nuclei and moderate cytoplasm. IHC confirmed strong nuclear positivity for TTF-1 and granular cytoplasmic staining for napsin A, supporting glandular differentiation (Fig. 1a–c).
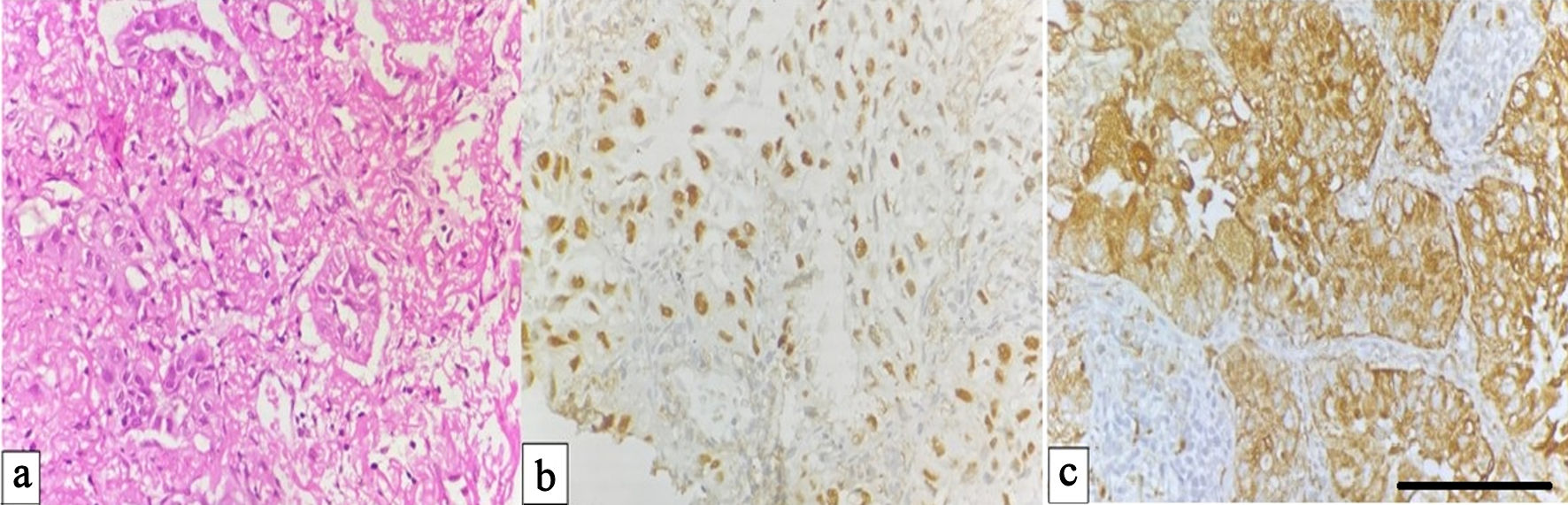 Click for large image | Figure 1. Adenocarcinoma of the lung. (a) Hematoxylin and eosin (H&E) section showing malignant gland-forming epithelial cells (× 400). (b) Immunohistochemical staining demonstrating nuclear positivity for TTF-1 in tumour cells (× 400). (c) Cytoplasmic staining for napsin A confirming adenocarcinomatous differentiation in tumor cells (× 400, scale bar = 50 µm). TTF-1: thyroid transcription factor-1. |
Immunohistochemical marker expression
A comprehensive IHC panel (TTF-1, napsin A, CK7, CK5/6, P63, synaptophysin, and chromogranin) was used to optimize tumor subclassification (Table 2). To further evaluate the diagnostic performance of the immunohistochemical panel, sensitivity and specificity were calculated for key lineage-specific markers. TTF-1 and napsin A demonstrated high sensitivity for adenocarcinoma (85.2% and 94.3%, respectively), with napsin A showing 100% specificity. For squamous differentiation, CK5/6 exhibited high sensitivity (92.3%) and specificity (98.6%), whereas P63 demonstrated lower sensitivity (61.5%) but retained high specificity (98.6%). Synaptophysin and chromogranin showed excellent performance in neuroendocrine tumors, with sensitivities of 96.1% and 100%, respectively, and both markers demonstrating 100% specificity (Table 3). Key observations included that TTF-1 (85%) and napsin A (94%) were highly sensitive for adenocarcinoma. CK5/6 (100%) and P63 (100%) were specific for squamous differentiation. Synaptophysin (96%) and chromogranin (100%) confirmed neuroendocrine origin. Vimentin, smooth muscle actin (SMA), and S-100 protein (S-100) showed focal positivity in carcinosarcoma, indicating epithelial–mesenchymal transition (Table 4).
 Click to view | Table 2. IHC Expression Pattern and Specificity (N = 151) |
 Click to view | Table 3. Diagnostic Performance of Key Immunohistochemical Markers |
 Click to view | Table 4. IHC Profiles of Metastatic Lesions to the Lung |
Metastatic lesions
Among the 20 metastatic cases, the lung served as a secondary site for breast carcinoma (10 cases), renal cell carcinoma (six cases), and oral squamous carcinoma (four cases). Breast metastases demonstrated positivity for ER, PR, HER-2, E-cadherin, and CK7. Renal metastases were positive for PAX-2, PAX-8, and CD10. Oral squamous metastases showed strong expression of CK5/6 and P63. All metastatic lesions were TTF-1 negative, confirming their extrapulmonary origin. The oral cavity metastases demonstrated sheets of keratinizing squamous cells with intercellular bridges and strong nuclear P63 positivity, confirming squamous lineage (Fig. 2a, b).
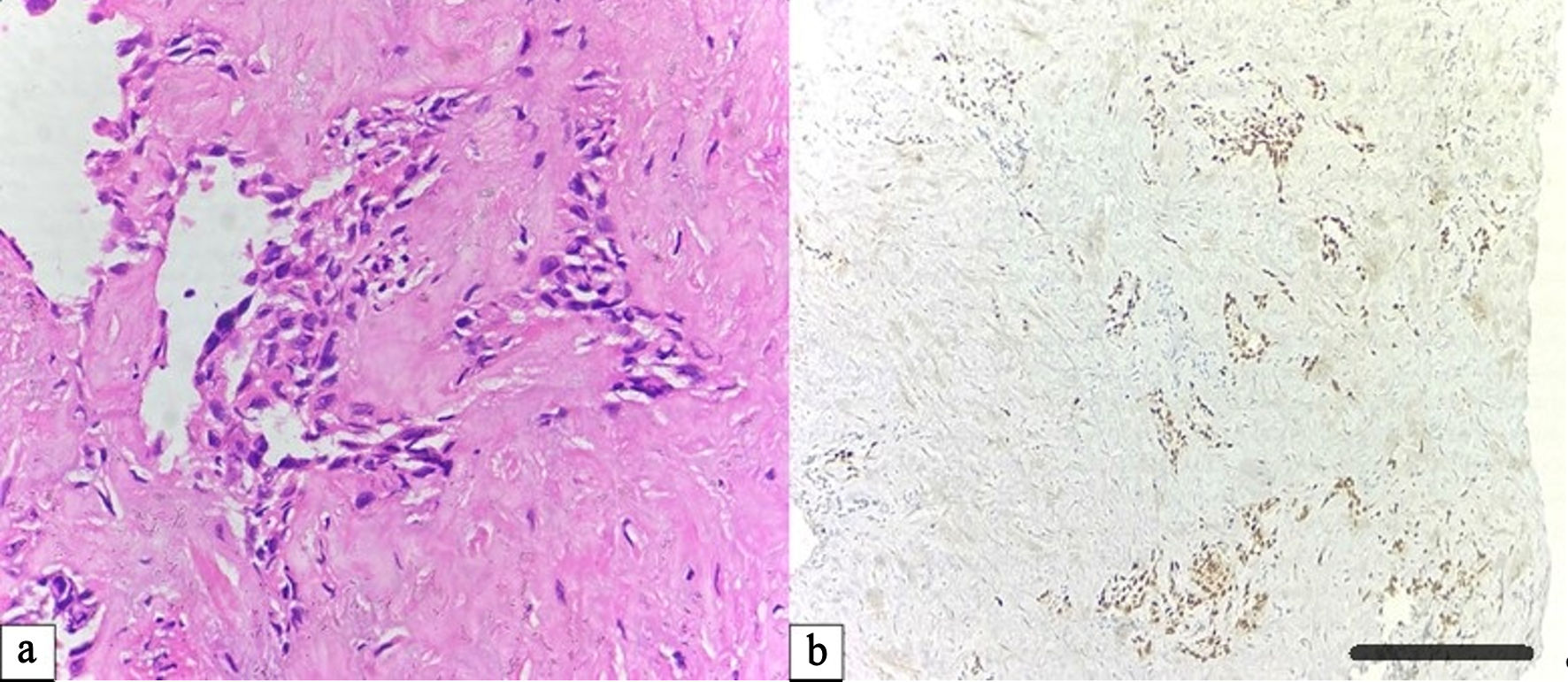 Click for large image | Figure 2. Pulmonary metastasis from oral squamous cell carcinoma. (a) Hematoxylin and eosin (H&E) section showing nests of keratinizing squamous cells with marked pleomorphism and intercellular bridges (× 400). (b) Tumor cells exhibit diffuse nuclear immunoreactivity for P63, supporting squamous origin (× 100, scale bar = 100 µm). |
Rare tumors
Two cases of carcinosarcoma displayed biphasic morphology with distinct epithelial and mesenchymal components. The adenocarcinoma component showed glandular differentiation and TTF-1 positivity, whereas the sarcomatous component demonstrated tumor giant cells, marked pleomorphism, and positivity for S-100, confirming heterologous mesenchymal differentiation (Fig. 3a–d).
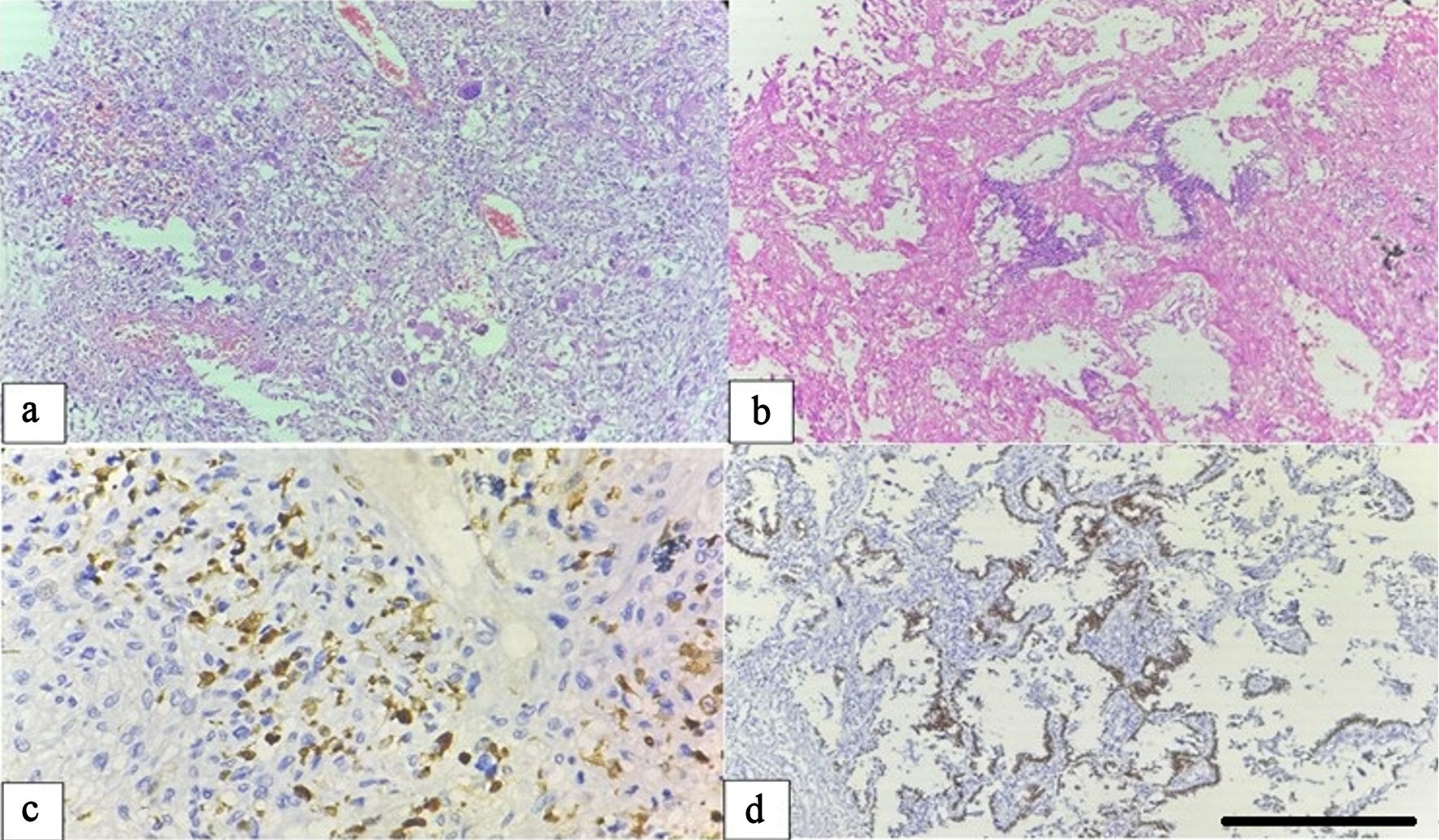 Click for large image | Figure 3. Carcinosarcoma of the lung. (a) Hematoxylin and eosin (H&E) section illustrating the sarcomatous component composed of pleomorphic spindle cells and tumor giant cells with prominent inflammatory infiltration (× 100). (b) Adenocarcinomatous component displaying glandular structures lined by atypical epithelial cells (× 100). (c) The sarcomatous component shows diffuse cytoplasmic positivity for S-100 protein (× 400). (d) The epithelial component exhibits nuclear TTF-1 positivity, confirming biphasic differentiation (× 100, scale bar = 100 µm). TTF-1: thyroid transcription factor-1. |
Correlation analysis between immunomarkers
Correlation analysis revealed strong linear associations between lineage-specific IHC markers (Table 4). TTF-1 vs napsin A in adenocarcinoma showed a strong positive correlation (r = 0.88, P < 0.001), confirming concordant expression in glandular tumors (Table 5). P63 vs CK5/6 in squamous and adenosquamous carcinomas demonstrated an excellent positive correlation (r = 0.93, P < 0.001), underscoring the robustness of this dual-marker panel for squamous differentiation. Synaptophysin vs chromogranin in neuroendocrine neoplasms displayed a highly significant correlation (r = 0.87, P < 0.001), reflecting consistent neuroendocrine lineage expression (Table 5). These correlations reinforce the diagnostic reliability and internal validity of the IHC panel and support its utility in the subclassification of lung tumors when only small tissue biopsies are available.
 Click to view | Table 5. Correlation Analysis Between Lineage-Specific Immunohistochemical Markers |
| Discussion | ▴Top |
This study provides a practice-oriented evaluation of immunohistochemical marker performance in the subclassification of lung tumors, with particular emphasis on real-world diagnostic scenarios involving limited biopsy material. Rather than introducing new biomarkers, our findings focus on validating the reliability, concordance, and clinical applicability of established markers when used as part of an integrated diagnostic panel [18, 23].
This approach aligns with contemporary diagnostic strategies, where IHC plays a central role in resolving morphologically ambiguous cases and guiding tumor classification in accordance with WHO recommendations [15].
The demographic pattern in this study, male predominance (2.1:1) and peak incidence in the fifth to seventh decades, mirrors previously reported epidemiologic distributions in both Asian and Western populations [5, 6]. Tobacco exposure remains a principal determinant, although the rising proportion of adenocarcinoma among non-smokers suggests a multifactorial etiology involving genetic susceptibility, pollution, and occupational hazards [24].
In the era of targeted and immune-based therapies, histopathological classification of lung cancer has become increasingly dependent on IHC profiling. Morphology alone may be insufficient, particularly in small biopsies or poorly differentiated tumors, necessitating marker-based lineage confirmation [16]. The present study underscores that the combination of TTF-1 and napsin A remains the cornerstone for identifying adenocarcinoma, demonstrating high sensitivity and specificity consistent with prior reports [18, 23]. TTF-1, a nuclear transcription factor expressed in alveolar epithelial cells, is indispensable for confirming pulmonary origin, while cytoplasmic napsin A provides complementary confirmation of glandular differentiation [25]. The strong correlation (r = 0.88, P < 0.001) between these markers in our series reinforces their combined diagnostic value and minimizes false negatives, especially when either antigen shows focal loss. The diagnostic performance analysis further supports the reliability of the selected IHC panel. In our study, TTF-1 and napsin A showed high sensitivity for adenocarcinoma, with napsin A achieving complete specificity, reinforcing its role as a robust marker of glandular differentiation. CK5/6 demonstrated excellent sensitivity and specificity for squamous differentiation, while P63, although highly specific, showed comparatively lower sensitivity, highlighting the importance of using combined marker panels. Similarly, synaptophysin and chromogranin exhibited near-perfect diagnostic performance for neuroendocrine tumors. These findings emphasize that a panel-based approach improves diagnostic confidence and minimizes misclassification in small biopsy specimens.
For squamous differentiation, the dual expression of P63 and CK5/6 showed complete concordance (r = 0.93, P < 0.001), in agreement with previous investigations by Kim et al (2013) and Gondha et al (2020) [18, 26]. P63, a p53-homologous transcription factor, identifies basal-type epithelial differentiation, whereas CK5/6 reflects keratinocyte-related cytokeratin expression. Their coexpression provides a highly reliable diagnostic framework for distinguishing squamous carcinoma from adenocarcinoma, particularly in mixed or adenosquamous lesions [26]. Moreover, the use of P40, a more specific ΔNp63 isoform, has been recommended in the latest WHO update, although P63 remains widely adopted in resource-limited settings [16].
In neuroendocrine neoplasms, synaptophysin and chromogranin demonstrated high and concordant positivity (r = 0.87, P < 0.001), confirming their utility as complementary neuroendocrine markers. This observation supports findings by Rudin et al (2021) and Raso et al (2021), emphasizing that dual-marker confirmation enhances diagnostic confidence and helps stratify neuroendocrine tumors ranging from typical carcinoids to small-cell carcinoma [13, 20]. Notably, the expression profile observed in this study mirrors the classical pattern described in large international cohorts, suggesting methodological consistency and biomarker robustness.
A key diagnostic challenge addressed in this study involves differentiating primary lung tumors from secondary metastatic deposits. Among our 20 metastatic cases, the absence of TTF-1 expression proved crucial for excluding primary origin. The integration of organ-specific markers such as ER/PR/HER-2 for breast, PAX-8/PAX-2/CD10 for renal, and CK5/6/P63 for oral cavity metastases provided precise site attribution. Similar marker-based diagnostic strategies have been validated by Vidarsdottir et al (2019) and Carney et al (2015), who emphasized the role of TTF-1 negativity in the recognition of extrapulmonary metastases [21, 22]. These findings underscore the importance of a systematic marker panel in avoiding diagnostic pitfalls, particularly in metastatic settings where treatment decisions hinge on accurate tumor origin. However, CK7 lacks specificity, as it is expressed across a wide range of epithelial malignancies; therefore, it should be interpreted in combination with other lineage-specific markers rather than as a standalone diagnostic indicator.
The correlation analysis conducted in this study provides essential quantitative validation of IHC marker performance. Strong positive correlations across adenocarcinoma, squamous, and neuroendocrine subsets confirmed the internal consistency of the staining protocol and the reproducibility of marker coexpression. This data-driven validation not only reinforces diagnostic accuracy but also supports the development of streamlined minimal marker panels for routine clinical application—particularly valuable in low-resource or high-throughput pathology laboratories [7]. Our findings are comparable to those of Bhatti et al, who reported adenocarcinoma as the predominant histotype (59%), followed by squamous and neuroendocrine carcinomas, and to Ericson Lindquist et al (2021), who demonstrated real-world accuracy exceeding 90% for IHC-based lung cancer diagnosis [6, 7]. The parallel trends in marker specificity—TTF-1/napsin A for adenocarcinoma and CK5/6/P63 for squamous carcinoma—reflect a global consensus in contemporary lung cancer pathology [8].
The integration of histopathology with a targeted IHC panel in this study enhanced diagnostic precision, particularly in small biopsy samples where morphology alone can be inconclusive. However, this study’s limitations include the absence of molecular profiling (e.g., epidermal growth factor receptor (EGFR), anaplastic lymphoma kinase (ALK), Kirsten rat sarcoma viral oncogene homolog (KRAS), or programmed death-ligand 1 (PD-L1)), which is now integral to personalized therapy. Future research incorporating molecular–IHC correlation would provide a more comprehensive diagnostic and prognostic framework [17, 27].
In the current era of precision oncology, molecular profiling has become an essential component of lung cancer diagnosis and management. Alterations in genes such as EGFR, ALK, and KRAS, as well as PD-L1 expression, are now routinely used to guide targeted and immunotherapy-based treatment strategies [11, 27]. While the present study focuses on immunohistochemical classification, IHC serves as a critical first step in tumor subtyping, thereby directing appropriate molecular testing. The integration of morphological, immunophenotypic, and molecular data provides a comprehensive diagnostic framework aligned with contemporary WHO recommendations [17].
An important strength of this study lies in its data-driven validation of marker concordance and diagnostic performance within a real-world cohort. The strong correlations observed between lineage-specific markers, together with high sensitivity and specificity values, demonstrate the internal consistency and robustness of the applied IHC panel. This is particularly relevant in routine pathology practice, where diagnostic decisions frequently rely on small biopsy specimens and optimized marker selection rather than extensive molecular profiling [10].
By quantitatively supporting marker reliability, our findings provide practical guidance for improving diagnostic confidence and reducing misclassification in daily clinical workflows, consistent with previous real-world studies demonstrating high diagnostic accuracy of IHC-based classification [7, 12].
Overall, this study validates the crucial diagnostic role of IHC in subclassifying lung tumors, differentiating primary from metastatic lesions, and providing lineage-specific insights that align with the WHO 2021 framework. The demonstrated marker correlations confirm the robustness and reproducibility of the applied panel. By integrating morphology with quantitative IHC data, this study supports a precision pathology approach that strengthens diagnostic reliability and informs personalized management strategies for patients with lung cancer.
Taken together, these findings reinforce the role of IHC not merely as a confirmatory tool but as a central, decision-guiding component in lung cancer diagnosis, particularly in biopsy-limited and resource-variable settings.
Conclusions
In conclusion, this study demonstrates that a focused immunohistochemical panel provides a robust and reproducible approach for lung tumor classification, particularly in small biopsy specimens where morphological assessment alone may be insufficient. The strong concordance observed among lineage-specific markers reinforces their diagnostic reliability, while integrating IHC with histopathology enables accurate distinction between primary and metastatic lesions. These findings highlight the continued relevance of IHC as a practical and clinically impactful tool in lung cancer diagnostics.
Acknowledgments
None to declare.
Financial Disclosure
None to declare.
Conflict of Interest
The authors have no relevant conflict of interest to disclose.
Informed Consent
A waiver of informed consent was granted by the Institutional Review Board, as the study utilized archived biopsy specimens and involved no direct patient interaction.
Author Contributions
Clinical data collection, interpretation, and analysis were performed by IT, AH, BN, SK, MA, HW, HN, AA, AF, RA, MK, and MAl. Methodology and investigation were conducted by AH, SK, AA, MK, and MAl. IT, HW, and HN performed biostatistical analysis and project administration. All authors critically reviewed and approved the final manuscript and are responsible for its content and similarity index.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding authors (MAl) and (AH) on reasonable request.
| References | ▴Top |
- Thandra KC, Barsouk A, Saginala K, Aluru JS, Barsouk A. Epidemiology of lung cancer. Contemp Oncol (Pozn). 2021;25(1):45-52.
doi pubmed - Zheng M. Classification and pathology of lung cancer. Surg Oncol Clin N Am. 2016;25(3):447-468.
doi pubmed - Coudray N, Ocampo PS, Sakellaropoulos T, Narula N, Snuderl M, Fenyo D, Moreira AL, et al. Classification and mutation prediction from non-small cell lung cancer histopathology images using deep learning. Nat Med. 2018;24(10):1559-1567.
doi pubmed - Krishnamurthy A, Vijayalakshmi R, Gadigi V, Ranganathan R, Sagar TG. The relevance of "Nonsmoking-associated lung cancer" in India: a single-centre experience. Indian J Cancer. 2012;49(1):82-88.
doi pubmed - Malik PS, Sharma MC, Mohanti BK, Shukla NK, Deo S, Mohan A, Kumar G, et al. Clinico-pathological profile of lung cancer at AIIMS: a changing paradigm in India. Asian Pac J Cancer Prev. 2013;14(1):489-494.
doi pubmed - Bhatti V, Kwatra KS, Puri S, Calton N. Histopathological spectrum and immunohistochemical profile of lung carcinomas: a 9-year study from a tertiary hospital in North India. Int J Appl Basic Med Res. 2019;9(3):169-175.
doi pubmed - Ericson Lindquist K, Gudinaviciene I, Mylona N, Urdar R, Lianou M, Darai-Ramqvist E, Haglund F, et al. Real-world diagnostic accuracy and use of immunohistochemical markers in lung cancer diagnostics. Biomolecules. 2021;11(11):1721.
doi pubmed - Domen A, Deben C, De Pauw I, Hermans C, Lambrechts H, Verswyvel J, Siozopoulou V, et al. Prognostic implications of cellular senescence in resected non-small cell lung cancer. Transl Lung Cancer Res. 2022;11(8):1526-1539.
doi pubmed - Passiglia F, Bertaglia V, Reale ML, et al. Significant breakthroughs in lung cancer adjuvant treatment: looking beyond the horizon. Cancer Treat Rev. 2021;101:102308.
- McLean AEB, Barnes DJ, Troy LK. Diagnosing lung cancer: the complexities of obtaining a tissue diagnosis in the era of minimally invasive and personalised medicine. J Clin Med. 2018;7(7):163.
doi pubmed - Ye X, Yang XZ, Carbone R, Barshack I, Katz RL. Diagnosis of non-small cell lung cancer via liquid biopsy, highlighting a fluorescence-in-situ-hybridisation circulating tumour cell approach. In: Strumfa I, Bahs G, editors. Pathology: from classics to innovations. London: IntechOpen; 2021.
- Sukswai N, Khoury JD. Immunohistochemistry innovations for diagnosis and tissue-based biomarker detection. Curr Hematol Malig Rep. 2019;14(5):368-375.
doi pubmed - Raso MG, Bota-Rabassedas N, Wistuba II. Pathology and classification of SCLC. Cancers (Basel). 2021;13(4):820.
doi pubmed - Whithaus K, Fukuoka J, Prihoda TJ, Jagirdar J. Evaluation of napsin A, cytokeratin 5/6, p63, and thyroid transcription factor 1 in adenocarcinoma versus squamous cell carcinoma of the lung. Arch Pathol Lab Med. 2012;136(2):155-162.
doi pubmed - Travis WD, Brambilla E, Nicholson AG, Yatabe Y, Austin JHM, Beasley MB, Chirieac LR, et al. The 2015 World Health Organization Classification of lung tumors: impact of genetic, clinical and radiologic advances since the 2004 classification. J Thorac Oncol. 2015;10(9):1243-1260.
doi pubmed - Thamtam V, Uppin S, Hui M, et al. Validation of the superiority of p40 over p63 in differentiating squamous cell carcinoma and adenocarcinoma of the lung. Indian J Med Paediatr Oncol. 2020;41:535-542.
- Liam CK, Mallawathantri S, Fong KM. Is tissue still the issue in detecting molecular alterations in lung cancer? Respirology. 2020;25(9):933-943.
doi pubmed - Kim MJ, Shin HC, Shin KC, Ro JY. Best immunohistochemical panel in distinguishing adenocarcinoma from squamous cell carcinoma of lung: tissue microarray assay in resected lung cancer specimens. Ann Diagn Pathol. 2013;17(1):85-90.
doi pubmed - Pelosi G, Rossi G, Bianchi F, Maisonneuve P, Galetta D, Sonzogni A, Veronesi G, et al. Immunhistochemistry by means of widely agreed-upon markers (cytokeratins 5/6 and 7, p63, thyroid transcription factor-1, and vimentin) on small biopsies of non-small cell lung cancer effectively parallels the corresponding profiling and eventual diagnoses on surgical specimens. J Thorac Oncol. 2011;6(6):1039-1049.
doi pubmed - Rudin CM, Brambilla E, Faivre-Finn C, Sage J. Small-cell lung cancer. Nat Rev Dis Primers. 2021;7(1):3.
doi pubmed - Vidarsdottir H, Tran L, Nodin B, Jirstrom K, Planck M, Jonsson P, Mattsson JSM, et al. Immunohistochemical profiles in primary lung cancers and epithelial pulmonary metastases. Hum Pathol. 2019;84:221-230.
doi pubmed - Carney JM, Kraynie AM, Roggli VL. Immunostaining in lung cancer for the clinician. Commonly used markers for differentiating primary and metastatic pulmonary tumors. Ann Am Thorac Soc. 2015;12(3):429-435.
doi pubmed - Zhao W, Wang H, Peng Y, Tian B, Peng L, Zhang DC. DeltaNp63, CK5/6, TTF-1 and napsin A, a reliable panel to subtype non-small cell lung cancer in biopsy specimens. Int J Clin Exp Pathol. 2014;7(7):4247-4253.
pubmed - Corrales L, Rosell R, Cardona AF, Martin C, Zatarain-Barron ZL, Arrieta O. Lung cancer in never smokers: the role of different risk factors other than tobacco smoking. Crit Rev Oncol Hematol. 2020;148:102895.
doi pubmed - Alekhya M, Rukmangadha N, Lakshmi AY, Manickavasagam M. Role of immunohistochemistry in the subtyping of non-small cell lung carcinoma on accurate cut lung biopsies. Ann Pathol Lab Med. 2018;5:447-455.
- Gondha S, Trivedi P, Patel N, Vora H. Role of immunohistochemistry in the subtyping of non-small cell lung carcinoma. Indian J Pathol Oncol. 2020;7:269-272.
- Masood A, Kancha RK, Subramanian J. Epidermal growth factor receptor (EGFR) tyrosine kinase inhibitors in non-small cell lung cancer harboring uncommon EGFR mutations: Focus on afatinib. Semin Oncol. 2019;46(3):271-283.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, including commercial use, provided the original work is properly cited.
World Journal of Oncology is published by Elmer Press Inc.