| World Journal of Oncology, ISSN 1920-4531 print, 1920-454X online, Open Access |
| Article copyright, the authors; Journal compilation copyright, World J Oncol and Elmer Press Inc |
| Journal website https://wjon.elmerpub.com |
Original Article
Volume 000, Number 000, April 2025, pages 000-000
Co-expression of HER2/EGFRvIII/CD44 and Claudin 18.2/CD109 as Novel Prognostic Indicators in Stomach Adenocarcinoma
Tina Al Janabya, Said Khelwattyb, Izhar Bagwana, b, Nima Abbassi-Ghadic, Helmout Modjtahedia, d
aDepartment of Biomolecular Science, School of Life Science, Pharmacy and Chemistry, Kingston University London, Kingston, UK
bBerkshire Surrey Pathology Services, Royal Surrey Hospital, Guildford, UK
cDepartment of Esophagogastric Surgery, Royal Surry Hospital, Guildford, UK
dCorresponding Author: Helmout Modjtahedi, School of Life Science, Pharmacy and Chemistry, Kingston University London, Kingston-upon-Thames, Surrey KT1 2EE, UK
Manuscript submitted February 5, 2025, accepted March 26, 2025, published online April 4, 2025
Short title: Prognostic of HER in Stomach Adenocarcinoma
doi: https://doi.org/10.14740/wjon2552
| Abstract | ▴Top |
Background: The heterogenous expression of human epidermal growth factor receptor (HER) family members may contribute to poor response to current therapies with HER inhibitors in cancer. This study aimed to explore the co-expression and prognostic significance of HER family members with epidermal growth factor receptor variant III (EGFRvIII), cluster of differentiation 44 (CD44), cluster of differentiation 109 (CD109), and claudin 18.2 (CLDN18.2) in patients with stomach cancer.
Methods: The relative expression and prognostic significance of these biomarkers at different cut-off values were determined in 78 patients with stomach adenocarcinoma by immunohistochemistry.
Results: Of the 78 cases, positive tumor staining was present for wild-type EGFR (13%), HER2 (82%), HER3 (9%), HER4 (33%), EGFRvIII (33%), CD44 (41%), CD109 (60%), and CLDN18.2 (40%). Furthermore, the expression of HER2 was accompanied with the co-expression of EGFR (9%), HER3 (8%), HER4 (27%), EGFRvIII (28%), CD44 (33%), CD109 (49%), and CLDN18.2 (32%). Interestingly, at the cut-off value ≥ 5% of tumor cells with positive staining, the co-expressions of HER2/EGFRvIII, EGFRvIII/CD44, and HER2/EGFRvIII/CD44 were associated with poor overall survival. Moreover, CLDN18.2 immunostaining of intensity of 3+, membranous expression of CD109, the co-expression of CD109/CLDN18.2 and CD109/EGFRvIII/CD44 were also associated with poorer overall survival and a higher risk of poor overall survival. All these remained as independent prognostic factors for survival in multivariate analysis.
Conclusion: This study provides first comprehensive analysis of the novel biomarker combinations that are significantly associated with overall survival. Co-expression of HER2 with EGFRvIII, CD44, and CD109, plus membranous CD109 and high-intensity CLDN18.2, independently predicted poor survival in stomach adenocarcinoma, highlighting their potential as prognostic biomarkers. These biomarker combinations may represent potential therapeutic targets for novel combination therapies, and future studies should investigate their predictive value for the response to therapy.
Keywords: Stomach cancer; Prognosis; EGFRvIII; CD44; Claudin 18.2; CD109
| Introduction | ▴Top |
The aberrant expression of human epidermal growth factor receptor (HER) family members has been reported in a number of human cancers and has been associated with tumor proliferation, progression, and metastasis. In some studies, it has also been associated with patients’ prognosis [1-4]. Of the HER family members, epidermal growth factor receptor (EGFR) and HER2 have become attractive targets for therapy with monoclonal antibodies and small tyrosine kinase inhibitors in several human malignancies including breast, non-small cell lung cancer (NSCLC), and stomach cancer [5-7]. Despite such advances, many patients do not benefit from therapy with the HER inhibitors alone, suggesting that the expression of other factors may contribute to the poor response or the development of resistance to treatment with various therapeutic agents. For example, the aberrant expression of cancer stem cells (CSCs) has been implicated in several cancers due to their role in cancer initiation, proliferation and progression, as well as their ability to self-renew and differentiate into various tumor cell types. Therefore, the presence of CSCs may contribute to the tumor heterogeneity and ultimately chemotherapy resistance as well as a poorer prognosis in cancer patients [8-10]. Indeed, in some studies, the presence of CSCs and the crosstalk between the HER family members have been associated with resistance to anti-HER2-targeted therapy in patients with breast and stomach cancers [11-14].
Of the CSCs, cluster of differentiation 44 (CD44) is a transmembrane glycoprotein receptor that binds to hyaluronic acid which cascades intracellular signaling that results in function such as cell adhesion, migration, and invasion [8, 15]. In other studies, the expression of CD109, which is a glycoprotein that is a member of the α2-macroglobulin/complement family and a co-receptor of transforming growth factor (TGF)-β, has been reported in several human malignancies and has been associated with poor prognosis [16-18]. In stomach cancer, the expression of CD109 has been shown to induce 5-flourouracil (5-FU) resistance in NCI-N87 cell line [19]. In addition, the interactions between CD109 and EGFR have been shown to promote cancer progression [20]. However, there are no studies investigating the co-expression and prognostic significance of CD109 and HER family members in patients with stomach cancer.
In recent years, the expression of claudin (CLDN)18.2 has been widely investigated as a potential therapeutic target for stomach cancer. CLDN18.2 is a highly selective gastric lineage marker that is expressed in short-lived differentiated cells of the stomach mucosa. However, during malignant transformation, the loss of cell polarity exposes the epitope of CLDN18.2, making it accessible to antibodies and a potential therapeutic target [21, 22]. In March 2024, the anti-CLDN18.2 antibody zolbetuximab was approved for the treatment of HER2-negative, CLDN18.2-positive stomach cancer and gastroesophageal cancer patients in Japan [23]. In October 2024, anti-CLDN18.2 mAb zolbetuximab was FDA approved as a first-line treatment for patients with locally advanced unresectable/metastatic HER2-negative gastric or gastroesophageal junction (GEJ) adenocarcinoma whose tumors are CLDN18.2-positive [24, 25]. The approval was based on the results obtained from SPOTLIGHT and GLOW clinical trials where the combination of zolbetuximab with mFOLFOX6 or CAPOX increased progression-free survival (PFS) by 1.94 and 1.41 months, respectively [26].
While the expression of individual HER family members and related biomarkers has been studied in stomach cancer, the complex interplay and co-expression of the entire HER family (including epidermal growth factor receptor variant III (EGFRvIII)), CD44, CD109, and CLDN18.2 remain largely unexplored. This study aimed to uniquely address this critical gap by examining the co-expression of all these biomarkers and providing a comprehensive analysis of their prognostic significance in stomach adenocarcinoma patients.
| Materials and Methods | ▴Top |
Patients
In this study, 78 stomach adenocarcinoma patients who underwent laparoscopic subtotal/total gastrostomy or open subtotal/total gastrostomy at the Royal Surry Hospital (Guildford, UK) between 2006 and 2019 were included. Ethical approval was obtained from the NHS Health Research Authority (HRA) and Health and Care Research Wales (HCRW) (IRAS approval ID: 277537) and as only archived tumor specimens were included, the ethics committee waived the need for patient consent and patient information were analyzed anonymously. Tumor blocks with insufficient tumor and no follow-up information were excluded from this study. Complete clinicopathological characteristics including gender, tumor stage, depth of invasion, and tumor grade were available for each patient. The median follow-up period for these patients was 5 years and the median age was 73.5 years which ranged between 29 and 93 years.
Immunohistochemistry
Serial sections of tumor specimens were cut from a batch of 78 paraffin-embedded blocks from stomach cancer patients. These sections were stained using primary antibodies as follows: mouse anti-EGFR (clone DAK-H1-WT, 1:100, Agilent Cat# M7298, RRID:AB_2286187), mouse anti-Her2/neu (clone 3B5, 1:200, Santa Cruz Biotechnology Cat# sc-33684, RRID:AB_627996), rabbit anti-HER3 (clone SP71, 1:50, Abcam Cat# ab93739, RRID:AB_10563976), mouse anti-HER4 (clone HFR1 1:100, Santa Cruz Biotechnology Cat# sc-53280, RRID:AB_629257), mouse anti-EGFRvIII (clone DH8.3, 1:500, Novus Cat# NBP2-50599, RRID:AB_3328879), mouse anti-CD44 (1:40, Agilent Cat# M7082, RRID:AB_2076596), and mouse anti-CD109 (30 µg/mL, KU42.33C, Kingston University, UK) [27], as described previously [17]. The staining for CLDN18.2 was conducted using rabbit anti-CLDN18.2 mAb (clone EPR19202, at 2 µg/mL, Abcam Cat# ab222512). Optimization of this antibody was conducted using and normal stomach tissue as positive control and negative control (no primary antibody) in the run. All slides were counterstained with hematoxylin, mounted, and hand cover slipped. All staining was carried out using the Ventana Discovery Ultra autostainer using the UltraView DAB kit (Roche, UK) as described previously [17], with exception of CLDN18.2 which was stained using the OptiView DAB Kit (Roche, UK).
Immunohistochemical scoring
All sections were scored depending on the percentage of tumor cells with positive immunostaining (≥ 5%, ≥ 10%, ≥ 20%, and ≥ 50%), the intensity of immunostaining (i.e., 0 = negative, 1+ = weak, 2+ = moderate, and 3+ = strong), and their subcellular location (i.e., membranous, cytoplasmic, and nuclear), as described previously [17]. Scoring was conducted by two independent observers (including a consultant histopathologist) who were blinded to all clinical information and any disparity in scoring was resolved by simultaneous reassessment of the staining by both observers.
Statistical analysis
The statistical analysis was conducted using the Statistical Package for Social Sciences software (IBM®, SPSS statistics version 28, RRID:SCR_002865, UK). The Chi-square test (Pearson’s Chi-square) and Fisher’s exact test were used to assess the correlation between the immunohistochemistry score and the patient clinicopathological data. Kaplan-Meier survival plots were used to determine the association between biomarker expression and overall survival and the difference between the individual groups was determined using a log-rank test. Cox regression univariate and multivariate analysis was conducted to confirm whether the association with the biomarker expression is an independent factor of overall survival. P ≤ 0.05 was considered statistically significant.
| Results | ▴Top |
Association between clinicopathological characteristics of stomach cancer patients and overall survival
The overall survival was determined by Kaplan-Meier curves and log-rank test [28]. The mean overall survival was 6.48 ± 0.65 years. The overall survival was found to be significantly poorer in patients with lymph node invasion (5.3 ± 0.7 versus 8.4 ± 1.8 years, P = 0.030) and in patients with perineural invasion (3.9 ± 0.7 vs. 7.3 ± 0.8 years, P = 0.019). Furthermore, patients with vascular invasion had reduced overall survival; however, it was not statistically significant (P > 0.05, Table 1).
 Click to view | Table 1. Patient Clinicopathological Characteristics and Association With Overall Survival Using Kaplan-Meier Analysis and Log-Rank Test in 78 Stomach Tumor Specimens |
Immunohistochemical expression of HER family members and EGFRvIII in stomach cancer patients
We examined the expression levels of the four HER family members (wild-type (wt)-EGFR, HER2, HER3, and HER4) and EGFRvIII in tumor specimens from 78 stomach adenocarcinoma patients. HER2 and HER4 expressions were substantially higher than wt-EGFR and HER3 (Table 2). Using a ≥ 5% tumor cell positivity cut-off, we detected wt-EGFR in 12.8% (10/78) of patients, with membranous and cytoplasmic staining exhibiting weak to strong intensity (Fig. 1a). In contrast, HER2 positivity was observed in 82% of patients, predominantly cytoplasmic (78%), with only 8% (6/78) showing strong intensity (Fig. 1b). HER3 was positive in 9% of cases, exhibiting membranous/cytoplasmic staining, while strong staining was rare (3%, 2/78) (Fig. 1c). HER4 positivity was found in 33% of cases, with primarily cytoplasmic/nuclear staining, and no cases exhibiting strong intensity (Fig. 1d). At the ≥ 5% cut-off, we observed EGFRvIII in 33% of cases, mostly cytoplasmic (22%) with weak intensity (Fig. 1e, Table 2). Increasing the cut-off to ≥ 50% reduced EGFRvIII positivity to 9% of cases (Table 2). Table 2 summarizes HER family immunostaining results across various positivity cut-offs.
 Click to view | Table 2. Immunohistochemical Expression/Co-Expression of HER Family Members, EGFRvIII, CD44, CD109, and CLDN18.2 |
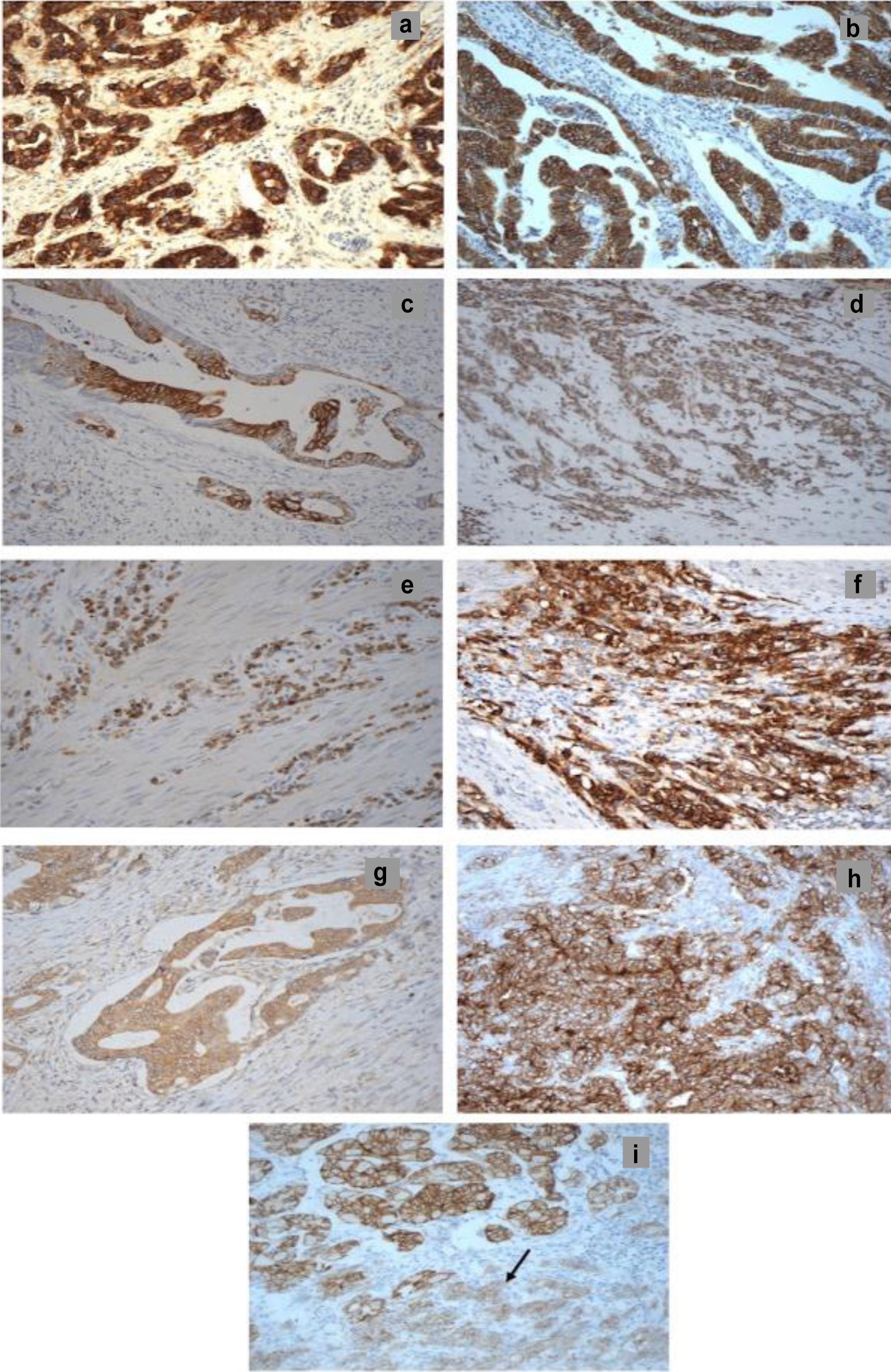 Click for large image | Figure 1. The immunohistochemical staining of HER family members, EGFRvIII, CD44, CD109, and CLDN18.2 in whole tumor specimens from patients with stomach adenocarcinoma (× 200 magnification). (a) EGFR 3+ membranous/cytoplasmic, (b) HER2 3+ membranous, (c) HER3 2+/3+ membranous, (d) HER4 1+ nuclear/cytoplasmic, (e) EGFRvIII 2+ cytoplasmic, (f) CD44 3+ membranous, (g) CD109 1+ membranous/cytoplasmic, (h) CLDN18.2 2+/3+ membranous, and (i) CLDN18.2 3+ in normal tissue and 2+ in tumor (arrow). CD44: cluster of differentiation 44; CD109: cluster of differentiation 109; CLDN18.2: claudin 18.2; EGFR: epidermal growth factor receptor; EGFRvIII: epidermal growth factor receptor variant III; HER2: human epidermal growth factor receptor. |
Immunohistochemical staining of CD44, CD109, and CLDN18.2 in stomach cancer
Using a ≥ 5% tumor cell positivity cut-off, we found CD44 to be positive in 41% of cases, localized to the cell membrane (Fig. 1f). Strong CD44 staining intensity was observed in 18% of cases. CD109 positivity was observed in 60% (47/78) of cases; however, staining intensity was predominantly weak, with only one patient showing moderate intensity. CD109 staining was membranous in 20% and cytoplasmic in 60% of cases (Fig. 1g). CLDN18.2 positivity (≥ 5% cut-off) was detected in 40% of cases, exclusively membranous. Strong staining intensity was present in 8% of cases, with weak and moderate intensity being observed in 24% and 18% of cases, respectively (Fig. 1h). We also noted CLDN18.2 expression in normal stomach tissue within the examined tumor blocks (Fig. 1i). Table 2 provides a summary of biomarker expression across various positivity cut-offs.
Co-expression of HER family with EGFRvIII, CD44, CD109, and CLDN18.2
As the heterogenous nature of human cancer and crosstalk between HER family members with other factors may play an important role in resistance to therapy, it is essential to determine the co-expression level of these biomarkers in stomach cancer. We found that at a cut-off value of ≥ 5% tumor cells with positive immunostaining, HER2 frequently co-expressed with wt-EGFR (9%) more than with HER3 (1%), HER4 (4%), or HER2/HER4 (1%) (Table 2 and Supplementary Material 1, wjon.elmerpub.com). Notably, the highest co-expression of two or more HER family members occurred between HER2 and HER4 (27%), while no patients exhibited co-expression of all four HER family members (Table 2 and Supplementary Material 1, wjon.elmerpub.com). Next, tumors from the same group of patients were examined for the co-expression of EGFRvIII with all four members of the HER family. Our findings indicated that tumors co-expressed EGFRvIII more commonly with HER2 (28%) than with wt-EGFR (5%), HER3 (4%), or HER4 (9%) (Table 2 and Supplementary Material 1, wjon.elmerpub.com).
For the first time in this study, we explored the co-expression of HER family members and EGFRvIII with other biomarkers, including CD44, CD109, and CLDN18.2. At the same cut-off value of ≥ 5%, we identified co-expression of wt-EGFR with CD44 (5%), CD109 (10%), and CLDN18.2 (3%). In contrast, we found that HER2 exhibited more frequent co-expression with CD44 (33%), CD109 (49%), and CLDN18.2 (32%). We further observed that 12% of cases showed co-expression of HER2 with EGFRvIII/CD44, 17% with EGFRvIII/CD109, 26% with CD44/CD109, and 15% with CD44/CD109/CLDN18.2. Additionally, in 31% of cases, we noted that CD44 co-expressed with CD109, in 19% with CLDN18.2, and in 18% with CD109/CLDN18.2. Lastly, we found that CD109 co-expressed with CLDN18.2 in 32% of the cases. These co-expression results, along with data from higher cut-off values that were less common, are summarized in Table 2 and Supplementary Material 1 (wjon.elmerpub.com).
Association between the clinicopathological characteristics and the expression of HER family members and EGFRvIII, CD44, CD109, and CLDN18.2
Following the determination of the expression of these biomarkers in the tumor specimens from 78 patients with stomach adenocarcinoma, a Fisher’s exact correlation test was performed to determine whether there was any significant association between the expression and co-expression of these biomarkers and the clinicopathological characteristics. The results of any significant associations between these biomarkers and clinicopathological parameters are presented in Table 3.
 Click to view | Table 3. Association Between Clinicopathological Characteristics and Protein Expression Using Chi-Square Test (FET) |
For example, at the cut-off value of ≥ 5% of the tumor cells with positive staining, the co-expression of HER2/CD109/CLDN18.2 was more common in patients over the age of 60 (P = 0.030). The expression of EGFRvIII was associated with depth of tumor and lymphatic invasion (Table 3). In addition, at cut-off value of ≥ 5% of the tumor cells, there was also a significant association between the co-expression of wt-EGFR/HER2, wt-EGFR/CD109, HER2/EGFRvIII and HER4/EGFRvIII, and lymphatic invasion. At the same cut-off, the co-expression of HER2/CD44/CLDN18.2 was found to be more common in diffuse/mixed type (P = 0.034, Table 3). The co-expression of HER2/CD109, at both cut values of ≥ 5% (P = 0.011) and ≥ 10% (P = 0.002) of the tumor cells with positive staining, was more common in higher grade tumors (Table 3). Finally, there was a statistically significant association between vascular invasion being present and the expression of EGFR at the cut-off value of ≥ 10% of the tumor cells with positive staining (P = 0.005, Table 3).
Association between the expression of HER family members, EGFRvIII, CD44, CD109, and CLDN18.2 and the overall survival in stomach cancer patients
There was no association between the expression of HER family members and overall survival in this study. Intriguingly, there was an association between the expression of EGFRvIII at intensity 1+ and poor overall survival in these patients (7.23 ± 0.79 vs. 4.48 ± 1.03 years, P = 0.037, Fig. 2a). The membranous expression of CD109 was also associated with poor overall survival in these patients (7.06 ± 0.75 vs. 3.93 ± 0.75 years, P = 0.028, Table 4, Fig. 2b). Moreover, patients with strong (intensity 3+) CLDN18.2 staining had poorer overall survival (6.83 ± 0.70 vs. 3.50 ± 1.50 years, P = 0.032, Fig. 2c).
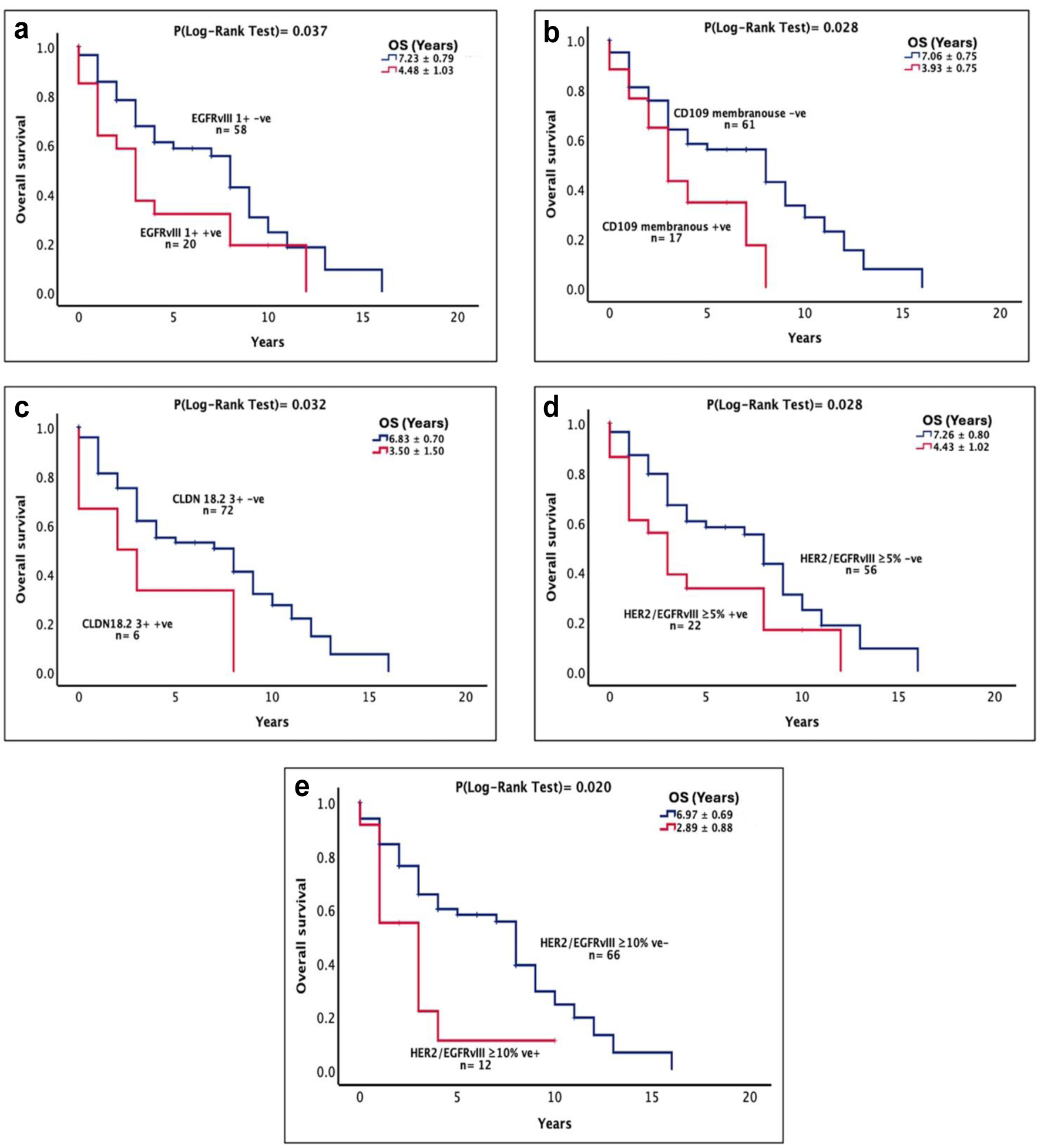 Click for large image | Figure 2. The prognostic significance and the association between the sub-categories of receptor expression and the OS of stomach cancer patients expressing EGFRvIII intensity 1+ (a), CD109 membranous expression (b), CLDN18.2 intensity 3+ (c), HER2/EGFRvIII at cut-off value of ≥ 5% (d), and HER2/EGFRvIII at cut-off value of ≥ 10% (e). A log-rank test value of P ≤ 0.05 was considered statistically significant. CD109: cluster of differentiation 109; CLDN18.2: claudin 18.2; EGFRvIII: epidermal growth factor receptor variant III; HER2: human epidermal growth factor receptor 2; OS: overall survival. |
 Click to view | Table 4. Association Between Expression of HER Family Members and Overall Survival in Univariate and Multivariate Analysis |
At cut-off values of ≥ 5% and ≥ 10% of tumor cells with positive immunostaining, the co-expression of HER2/EGFRvIII was associated with poor overall survival in these patients (Fig. 2d, e). Interestingly, at cut-off value of ≥ 5% of tumor cells with positive immunostaining, the co-expressions of HER2/EGFRvIII/CD44 (6.93 ± 0.69 vs. 2.33 ± 0.56 years, P = 0.011, Fig. 3a), EGFRvIII/CD44 (7.02 ± 0.69 vs. 2.17 ± 0.52 years, P = 0.003, Fig. 3b), and EGFRvIII/CD44/CD109 (7.01 ± 0.68 vs. 1.65 ± 0.50 years, P = 0.0001, Fig. 3c) were all associated with poorer survival outcome in these patients (Table 4). Furthermore, using univariate analysis, apart from EGFRvIII and CLDN18.2 intensity 1+, patients with CD109 membranous staining, CLDN18.2 intensity 3+, HER2/EGFRvIII/CD44, and EGFRvIII/CD44 co-expression were at a higher risk of poorer overall survival, which remained as independent prognostic factors in multivariate analysis (Table 4). Moreover, at the cut-off value ≥ 5% of tumor cells with positive immunostaining, patients with co-expression of EGFRvIII/CD44/CD109 had a 4.2-fold increased risk of poorer overall survival (P = 0.0009), which remained an independent prognostic factor in multivariate analysis (Table 4, P = 0.0009). Finally at the same cut-off value, patients with co-expression of CD109/CLDN18.2 had poor overall survival (7.01 ± 0.74 vs. 3.99 ± 0.97 years, P = 0.021, Fig. 3d) which also remained significant in multivariate analysis (P = 0.049).
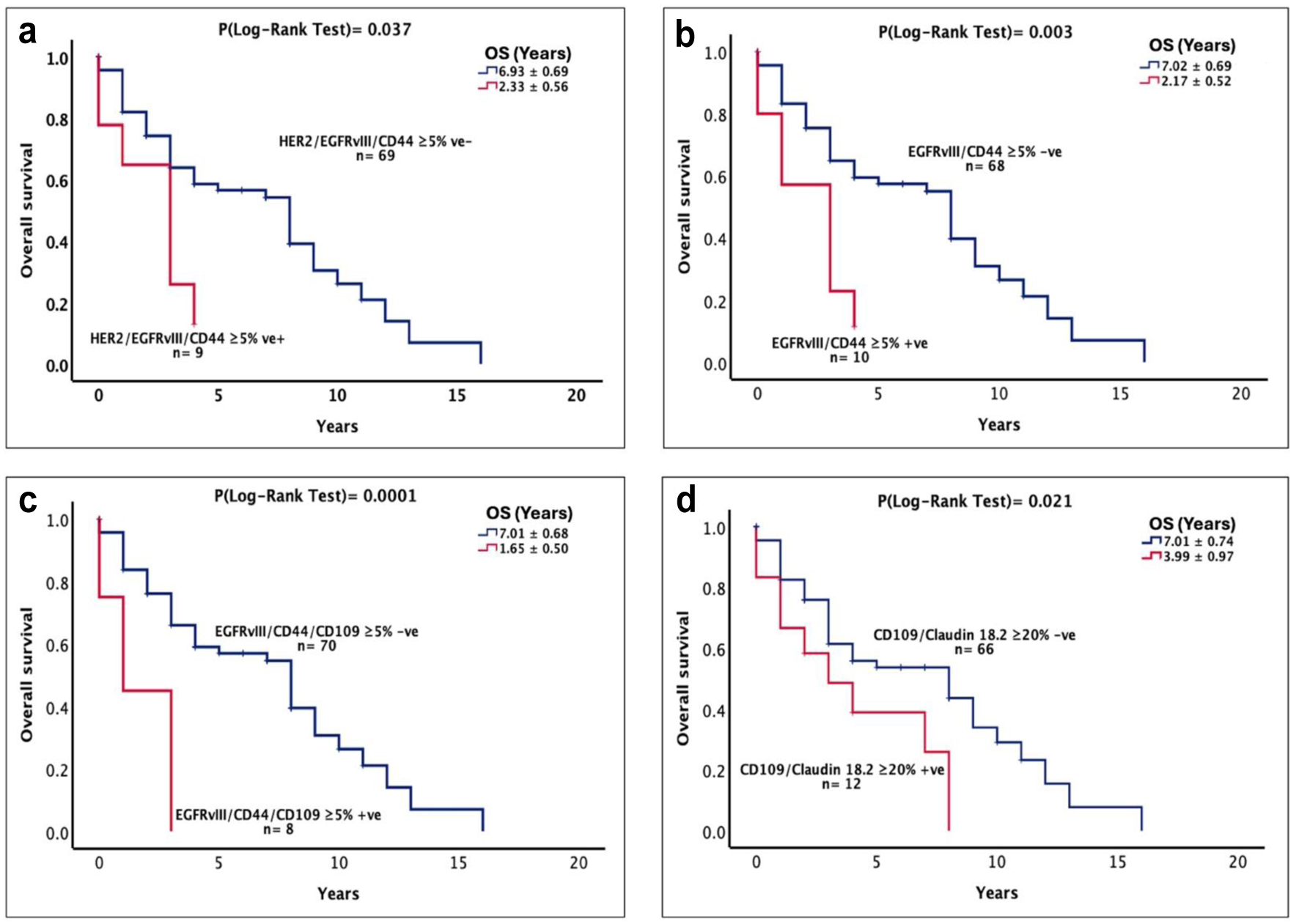 Click for large image | Figure 3. The prognostic significance and the association between the sub-categories of receptor expression and the OS of stomach cancer patients expressing HER2/EGFRvIII/CD44 at cut-off value of ≥ 5% (a), EGFRvIII/CD44 at cut-off value of ≥ 5% (b), EGFRvIII/CD44/CD109 at cut-off value of ≥ 5% (c), and CD109/CLDN18.2 at a cut-off value of ≥ 20% (d) in patients with stomach cancer. A log-rank test value of P ≤ 0.05 was considered statistically significant. CD44: cluster of differentiation 44; CLDN18.2: claudin 18.2; EGFRvIII: epidermal growth factor receptor variant III; HER2: human epidermal growth factor receptor 2; OS: overall survival. |
| Discussion | ▴Top |
Stomach cancer is one of the leading causes of cancer death worldwide and as patients are frequently diagnosed at an advanced stage, the 5-year survival rate is around 7.0% [29, 30]. Increased expression of HER family members has been reported in several human cancers including stomach cancer and in some cases associated with poor prognosis and they can be an independent prognostic factor of survival [3, 17, 31]. Of various types of EGFR mutations, the expression of EGFRvIII, a constitutively active, and ligand-independent receptor, has been reported in several cancers including glioblastoma and NSCLC [32, 33]; however, there are no studies investigating the co-expression level of all members of the HER family and EGFRvIII in patients with stomach cancer.
In this study, at the cut-off value of ≥ 5% of tumor cells with positive staining, the expression of wt-EGFR, HER2, HER3, and HER4 was present in 13%, 82%, 9%, and 33% of the cases examined, respectively (Table 2). However, the cellular location of EGFR was mainly cytoplasmic (12.8%) and only 3.8% of the cases had membranous expression of EGFR. The majority of studies investigating EGFR expression in stomach cancer have only focused on the membranous expression of EGFR, and did not report cytoplasmic expression of EGFR [31, 34, 35]. However, in other cancers such as pancreatic cancer and oral squamous cell carcinoma, cytoplasmic expression of EGFR has shown to be associated with disease aggressiveness and shorter overall survival [36, 37]. In this study, EGFR cytoplasmic expression was associated with lymphatic invasion but there was no statistically significant association between the expression of cytoplasmic EGFR and overall survival. In literature, there is no general agreement on the prognostic significance of EGFR in stomach cancer. In some retrospective studies, the EGFR expression was associated with poor survival [38-40], while other studies suggest that EGFR expression was not associated with poor survival [31, 34, 41]. The use of different antibodies (i.e., antibodies which recognize both wt-EGFR and EGFRvIII), different incubation times, sample size, and scoring methods could have contributed to the conflicting data on the prognostic significance of EGFR expression in stomach cancer patients. To date, clinical trials with anti-EGFR therapy have failed to show clinical efficacy in stomach cancer patients. Therefore, it is important to take into consideration the cellular location of EGFR, its phosphorylation status, and their prognostic significance when treating with various types of HER inhibitors targeting one or more members of the HER family and when used in combination with other target agents, which warrants further investigations in a larger group of patients [42-44].
In contrast to the wt-EGFR, EGFRvIII is a type III deletion-mutant receptor that is characterized by being constitutively active [45]. In the present study, the expression of EGFRvIII-positive cases (33.3%) was higher than the wt-EGFR (12.8%) and it was also mostly found in the cytoplasm of tumors (Table 2). In addition, the expression of EGFRvIII was associated with vascular and lymphatic invasion (Table 3). Interestingly, the expression of EGFRvIII intensity 1+ was significantly associated with poorer overall survival in this group of patients (P = 0.037, Fig. 2a). However, it did not remain as an independent prognostic factor in multivariate analysis (Table 4). The expression of EGFRvIII is observed in glioblastoma [46], head and neck cancer [47], and more recently in patients with hepatocellular carcinoma [48]. EGFRvIII is almost exclusively present with EGFR amplification in glioblastoma [32, 49]. However, in head and neck squamous cell carcinoma, EGFRvIII expression did not correlate with EGFR amplification, suggesting an alternative mechanism of EGFRvIII generation [47]. However, this is the first study examining the expression level and prognostic significance of EGFRvIII in patients with stomach cancer. Further investigation, using a larger group of patients, is warranted to confirm the relative expression, prognostic significance, and predictive value of EGFRvIII in patients with stomach cancer, and its potential as a target for therapy with anti-EGFRvIII specific antibodies and other inhibitors [50].
The expression of HER2 in this group of patients was higher (i.e., 82%), and with cellular location of HER2 staining being cytoplasmic and membranous in 78% and 13% of the cases examined, respectively (Table 2). In literature, the expression of HER2 varies between 8% and 40% of the cases examined, and usually scoring contained only the membranous expression of HER2. In one study, Ghaderi and colleagues showed HER2 to be expressed both in the membrane and cytoplasm in 16% of stomach tumor specimens using rat mAb IC12 [51]. Using HER2 mouse IgG clone CB11, Tewari and colleagues reported the membranous expression of HER2 in 21% of stomach cancer patients [52]. Furthermore, Ugras and colleagues used anti-HER2 mAb clone e2-4001-3B5 and found a membranous expression of HER2 in 40% of 56 patients with primary stomach adenocarcinoma [53]. Although HER2 was highly expressed in this group of patients, no association was found between the expression and overall survival. Similarly, several other studies have reported that HER2 is not a prognostic factor for overall survival [54-56]. Although data on the expression level and prognostic significance of HER2 remain unclear, HER2 expression or gene amplification is used as a biomarker for targeted therapy with the mAb trastuzumab and antibody-drug conjugate trastuzumab-deruxtecan in HER2-positive stomach/GEJ adenocarcinoma patients with unresectable/metastatic disease [6, 57]. In this study, HER2 expression was found to be high but membranous expression of HER2, which is a target antigen for anti-HER2 therapy, was present in 10% of the cases examined. Therefore, further investigation is warranted to determine the prognostic significance and predictive value of both cytoplasmic and membranous HER2 for the targeted therapy with HER2 inhibitors in patients with stomach cancer.
The crosstalk between the HER family and CSCs has been proven to promote tumorgenicity, migration, and metastasis in several human cancers [20, 58-60]. Therefore, in this study, we investigated the co-expression of HER family members with CD109, CD44, and CLDN18.2 for the first time, as well as their association with overall survival in patients (Table 2 and Fig. 2). In the present study, at cut-off values ≥ 5% of tumor cells, the co-expression of HER2/EGFRvIII was observed in 28% of cases examined and this was associated with a poor overall survival (Fig. 2d). Interestingly, the co-expression of HER2/EGFRvIII in ≥ 10% of tumor cells also had a poorer overall survival (Fig. 2e). We found that the co-expressions of HER2/EGFRvIII in ≥ 5% and ≥ 10% of tumor cells were independent prognostic factors for poor overall survival (Table 4). Yu and colleagues found that EGFRvIII and HER2 are co-expressed in 40% of primary breast cancer tumors and showed that this co-expression promoted tumorgenicity in transfected xenografts [61]. Moreover, in 32% of the patients, we found co-expression of HER2 with CLDN18.2 which was associated with increased age at both cut-off values of ≥ 5% and ≥ 10% of tumor cells (Table 3). In stomach cancer, CLDN18.2-positive tumors are usually HER2-negative. In agreement with our findings, results from the FAST trial showed that 14% (13/94) co-expressed CLDN18.2 with HER2 [62]. In another study, CLDN18.2 was found to be co-expressed with HER2 in 12% (10/83) of patients [63]. Furthermore, Sheng and colleagues observed increased expression of CLDN18.2 post-trastuzumab treatment, suggesting an association between trastuzumab resistance and upregulation of CLDN18.2 [64]. To our knowledge, this is the first study showing that co-expression of HER2 with CLDN18.2 or EGFRvIII is associated with increased age and poor survival in patients with stomach cancer, respectively. Therefore, further investigation should be conducted on the therapeutic potential of co-targeting such biomarkers in a larger cohort of patients with the stomach cancer and their predictive values for the response to therapy.
As mentioned previously, the expression of CD109 has been associated with poor overall survival and chemotherapy resistance in cancer [65, 66]. However, there were no studies examining the expression of CD109 in stomach cancer patients. In this study, the expression of CD109 was predominantly present in the cytoplasm (60%) of the tumor cases examined (Table 2). Furthermore, the membranous expression of CD109 was found to be associated with a poorer overall survival and had a two-fold increased risk of a poorer overall survival, which remained as a poor prognostic indicator of survival in multivariate analysis (Table 4). Khan and colleagues observed CD109 expression in 56% of whole tumor specimens of patients with pancreatic cancer. They also found that the expression of CD109 in > 50% of tumor cells was associated with a poorer overall survival. However, in multivariate analysis, it was not a prognostic indicator of overall survival [17]. Interestingly, in squamous cell carcinoma, CD109 was shown to promote tumorgenicity via heterodimerization with EGFR leading to the stabilization of EGFR levels and this dimerization regulates the cellular stemness through EGFR/AKT signaling [20]. The result of our study suggests that the expression of CD109 is high in patients with stomach cancer and has co-expression with wt-EGFR, HER2, HER3, and HER4. These results support further research on the co-expression and prognostic significance, and predictive value of such biomarkers in patients with stomach cancer as well as the therapeutic potential of the HER inhibitors when used in combination with anti-CD109 antibodies in stomach cancer [67].
Interestingly, in this study, patients with co-expression of CD109/CLDN18.2 in ≥ 20% of tumor cells had a significantly lower overall survival (7.01 ± 0.74 vs. 3.99 ± 0.97 years) and a 2.16 increased risk of poor overall survival which remained an independent prognostic factor (Fig. 3d and Table 4). As this is the first study on the co-expression of CD109 and CLDN18.2 in patients with stomach cancer, further study, involving a larger group of patients, is warranted, in particular to investigate the therapeutic potential of co-targeting CLDN18.2 with HER2 or CD109 in patients with stomach cancer which co-expresses such antigens and their predictive value for the response to such therapeutic interventions.
In this study, the expression of CD44 was primarily membranous and was present in 41% of tumor specimens examined (Table 2). There was no association between CD44 expression and clinicopathological characteristics or overall survival. Using the same Novocastra anti-CD44 antibody, Kodama et al and Wakamatsu et al found the expression of CD44 to be present in 10.6% (positivity defined as > 5% of cancer cells positively stained) and 62% (positivity defined as ≥ 10% of positive tumor cells) of stomach cancer patients, respectively. In addition, they found CD44 expression to be an independent prognostic factor of survival in patients with stomach cancer. Although these studies used the same antibody, the expression pattern varied between these studies and this could be due to the differences in scoring methods and experimental design [8, 68]. Further investigations are warranted for the prognostic significance of CD44 in stomach cancer patients.
At the cut-off value of ≥ 5% of tumor cells with positive staining, the co-expression of EGFRvIII and CD44 was present in 19.2% of the cases examined, associated with higher risk of poor overall survival in these patients and remained as an independent prognostic factor of poor overall survival (Fig. 3b, Table 4). In another study involving colorectal cancer patients, Khelwatty and colleagues also showed that the co-expression of EGFRvIII/CD44 was associated with shorter overall survival and remained an independent prognostic factor in such patients [69]. Finally, of other co-expressions of various biomarkers, the co-expression of EGFRvIII/CD44/CD109 in ≥ 5% of tumor cells had a worse overall survival compared to patients with negative co-expression (7.01 ± 0.68 vs. 1.65 ± 0.5, P = 0.0001, Fig. 3c). Moreover, these patients had a 4.2-fold higher risk of poor overall survival which remained as an independent prognostic factor when adjusted for multivariate analysis (P = 0.0009, Table 4). These results suggest that the co-expression of EGFRvIII with CD44 and CD109 can be used as prognostic indicators of survival.
Finally, CLDN18.2 expression has been shown to be prevalent in stomach cancer and became a targetable biomarker for the treatment of HER-negative locally advanced unresectable or metastatic HER2-negative gastric/GEJ adenocarcinoma [24, 25]. In this study, the presence of CLDN18.2 in normal stomach mucosa was seen in the majority of patients (Fig. 1i). There is evidence suggesting that CLDN18.2 expression in normal cells is contained in tight junction supramolecular complexes of stomach mucosal cells, resulting in its epitope to be inaccessible to intravenous antibodies [21, 70]. Furthermore, at the cut-off value of ≥ 5% of positive tumor cells, CLDN18.2 expression was present in 40% with subcellular location in the membrane (Table 2, Fig. 1h). In addition, CLDN18.2 in ≥ 20% of tumor cells was associated with perineural invasion (P = 0.0043, Table 3). A study done by Jun and colleagues found that CLDN18.2 expression had an inverse correlation with perineural invasion while other studies found no correlation [71-73]. Interestingly in cholangiocarcinoma, CLDN18.2 expression was significantly associated with perineural invasion and aggressive disease [74]. Kaplan-Meier analysis revealed that patients with CLDN18.2 intensity 3+ had a lower overall survival compared to CLDN18.2-negative patients (6.83 ± 0.70 vs. 3.50 ± 1.50 years, P = 0.032, Fig. 2c) and this remained as an independent prognostic factor (Table 4). While CLDN18.2 positivity was associated with poor overall survival [75, 76], other studies found no significant association between CLDN18.2 and overall survival [72, 73, 77, 78]. In spite of this, clinical trials have investigated the clinical efficacy of targeting CLDN18.2 with zolbetuximab, an anti-CLDN18.2 antibody. The results obtained from this study suggest that CLDN18.2 expression of strong intensity is present, and its expression can lead to tumor invasion and poor overall survival. Further research is warranted to determine the relationship of CLDN18.2 expression with clinicopathological features and overall survival. Moreover, at the cut-off of ≥ 5%, ≥ 10%, and ≥ 20% of tumor cells with positive staining, 32%, 24%, and 17% of cases had co-expression of HER2 with CLDN18.2, respectively (Table 2), supporting the need for investigating the therapeutic application of co-targeting of HER2 and CLDN18.2 in tumors co-expressing these two biomarkers.
In summary, this study provides the first comprehensive analysis of the co-expression of HER family members (EGFR, HER2, HER3, HER4, and EGFRvIII), CD44, CD109, and CLDN18.2 in a cohort of 78 stomach adenocarcinoma patients. Our findings reveal significant associations between specific co-expression patterns of these biomarkers and overall survival. Notably, the co-expression of HER2 with EGFRvIII, CD44, and CD109, as well as the membranous expression of CD109 and high-intensity CLDN18.2 staining, independently predicted poorer overall survival. These results highlight the importance of the co-expression and prognostic significance of such biomarkers in patients with stomach cancer. Furthermore, these biomarker combinations represent potential therapeutic targets for novel combination therapies. Future studies should investigate the predictive value and the efficacy of targeted agents against these specific molecular profiles to improve treatment response and potentially overcome resistance mechanisms.
| Supplementary Material | ▴Top |
Suppl 1. Co-expression of HER family members, EGFRvIII, CD44, and CD109 in stomach cancer tumor samples at different cut-off values.
Acknowledgments
We would like to acknowledge and thank the Norwegian Student Fund Lanekassen for providing the PhD scholarship.
Financial Disclosure
None to declare.
Conflict of Interest
The authors have no conflict of interest.
Informed Consent
Not applicable.
Author Contributions
HM is TAL’s Director of Studies, and provided study concept, design, data analysis and critical revision of the manuscript. TAL performed all the experiments, data analysis, drafting of manuscript and immunohistochemistry scoring. SK and IB are the co-supervisors on the project. SK helped with the training of various techniques and data analysis while IB did the immunohistochemistry scoring and patient information. NAG provided patient selection and follow-up information. All authors read and approved the final manuscript.
Data Availability
The authors declare that data supporting the findings of this study are available within the article and supplementary information file.
Abbreviations
CD44: cluster of differentiation 44; CD109: cluster of differentiation 109; CLDN18.2: claudin 18.2; CSC: cancer stem cell; EGFR: epidermal growth factor receptor; EGFRvIII: epidermal growth factor receptor variant III; FET: Fisher’s exact t-test; HER2: human epidermal growth factor receptor 2
| References | ▴Top |
- Khelwatty SA, Essapen S, Bagwan I, Green M, Seddon AM, Modjtahedi H. Co-expression of HER family members in patients with Dukes' C and D colon cancer and their impacts on patient prognosis and survival. PLoS One. 2014;9(3):e91139.
doi pubmed - Li Q, Zhang L, Li X, Yan H, Yang L, Li Y, Li T, et al. The prognostic significance of human epidermal growth factor receptor family protein expression in operable pancreatic cancer : HER1-4 protein expression and prognosis in pancreatic cancer. BMC Cancer. 2016;16(1):910.
doi pubmed - Wang SL, Zhong GX, Wang XW, Yu FQ, Weng DF, Wang XX, Lin JH. Prognostic significance of the expression of HER family members in primary osteosarcoma. Oncol Lett. 2018;16(2):2185-2194.
doi pubmed - Arienti C, Pignatta S, Tesei A. Epidermal growth factor receptor family and its role in gastric cancer. Front Oncol. 2019;9:1308.
doi pubmed - Neal JW. The SATURN trial: the value of maintenance erlotinib in patients with non-small-cell lung cancer. Future Oncol. 2010;6(12):1827-1832.
doi pubmed - Bang YJ, Van Cutsem E, Feyereislova A, Chung HC, Shen L, Sawaki A, Lordick F, et al. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial. Lancet. 2010;376(9742):687-697.
doi pubmed - Dawood S, Broglio K, Buzdar AU, Hortobagyi GN, Giordano SH. Prognosis of women with metastatic breast cancer by HER2 status and trastuzumab treatment: an institutional-based review. J Clin Oncol. 2010;28(1):92-98.
doi pubmed - Wakamatsu Y, Sakamoto N, Oo HZ, Naito Y, Uraoka N, Anami K, Sentani K, et al. Expression of cancer stem cell markers ALDH1, CD44 and CD133 in primary tumor and lymph node metastasis of gastric cancer. Pathol Int. 2012;62(2):112-119.
doi pubmed - Jung WY, Kang Y, Lee H, Mok YJ, Kim HK, Kim A, Kim BH. Expression of moesin and CD44 is associated with poor prognosis in gastric adenocarcinoma. Histopathology. 2013;63(4):474-481.
doi pubmed - Prasetyanti PR, Medema JP. Intra-tumor heterogeneity from a cancer stem cell perspective. Mol Cancer. 2017;16(1):41.
doi pubmed - Ritter CA, Perez-Torres M, Rinehart C, Guix M, Dugger T, Engelman JA, Arteaga CL. Human breast cancer cells selected for resistance to trastuzumab in vivo overexpress epidermal growth factor receptor and ErbB ligands and remain dependent on the ErbB receptor network. Clin Cancer Res. 2007;13(16):4909-4919.
doi pubmed - Boulbes DR, Chauhan GB, Jin Q, Bartholomeusz C, Esteva FJ. CD44 expression contributes to trastuzumab resistance in HER2-positive breast cancer cells. Breast Cancer Res Treat. 2015;151(3):501-513.
doi pubmed - Li X, Xu Y, Ding Y, Li C, Zhao H, Wang J, Meng S. Posttranscriptional upregulation of HER3 by HER2 mRNA induces trastuzumab resistance in breast cancer. Mol Cancer. 2018;17(1):113.
doi pubmed - Zhang M, Li B, Liao H, Chen Z, Huang W, Yang J, Ge S, et al. Targeting HER3 or MEK overcomes acquired Trastuzumab resistance in HER2-positive gastric cancer-derived xenograft. Cell Death Discov. 2022;8(1):478.
doi pubmed - Sneath RJ, Mangham DC. The normal structure and function of CD44 and its role in neoplasia. Mol Pathol. 1998;51(4):191-200.
doi pubmed - Tsutsumi S, Momiyama K, Ichinoe M, Kato T, Mogi S, Miyamoto S, Murakumo Y, et al. The significance of CD109 expression in oropharyngeal squamous cell carcinoma. Anticancer Res. 2022;42(4):2061-2070.
doi pubmed - Khan T, Seddon AM, Khelwatty SA, Dalgleish A, Bagwan I, Mudan S, et al. The Co-expression of HER family members and CD109 is common in pancreatic cancer. Medical Research Archives. 2023;11(11).
- Mori N, Esaki N, Shimoyama Y, Shiraki Y, Asai N, Sakai T, Nishida Y, et al. Significance of expression of CD109 in osteosarcoma and its involvement in tumor progression via BMP signaling. Pathol Res Pract. 2023;245:154443.
doi pubmed - Zhou F, Wang L, Ge H, Zhang D, Wang W. H3K27 acetylation activated-CD109 evokes 5-fluorouracil resistance in gastric cancer via the JNK/MAPK signaling pathway. Environ Toxicol. 2023;38(12):2857-2866.
doi pubmed - Zhou S, Hassan A, Kungyal T, Tabaries S, Luna J, Siegel PM, Philip A. CD109 Is a Critical Determinant of EGFR Expression and Signaling, and Tumorigenicity in Squamous Cell Carcinoma Cells. Cancers (Basel). 2022;14(15).
doi pubmed - Sahin U, Koslowski M, Dhaene K, Usener D, Brandenburg G, Seitz G, Huber C, et al. Claudin-18 splice variant 2 is a pan-cancer target suitable for therapeutic antibody development. Clin Cancer Res. 2008;14(23):7624-7634.
doi pubmed - Angerilli V, Ghelardi F, Nappo F, Grillo F, Parente P, Lonardi S, Luchini C, et al. Claudin-18.2 testing and its impact in the therapeutic management of patients with gastric and gastroesophageal adenocarcinomas: A literature review with expert opinion. Pathol Res Pract. 2024;254:155145.
doi pubmed - Keam SJ. Zolbetuximab: First Approval. Drugs. 2024;84(8):977-983.
doi pubmed - Shah MA, Shitara K, Ajani JA, Bang YJ, Enzinger P, Ilson D, Lordick F, et al. Zolbetuximab plus CAPOX in CLDN18.2-positive gastric or gastroesophageal junction adenocarcinoma: the randomized, phase 3 GLOW trial. Nat Med. 2023;29(8):2133-2141.
doi pubmed - Shitara K, Lordick F, Bang YJ, Enzinger P, Ilson D, Shah MA, Van Cutsem E, et al. Zolbetuximab plus mFOLFOX6 in patients with CLDN18.2-positive, HER2-negative, untreated, locally advanced unresectable or metastatic gastric or gastro-oesophageal junction adenocarcinoma (SPOTLIGHT): a multicentre, randomised, double-blind, phase 3 trial. Lancet. 2023;401(10389):1655-1668.
doi pubmed - Administration FD. FDA approves zolbetuximab-clzb with chemotherapy for gastric or gastroesophageal junction adenocarcinoma. Food & Drug Administration. 2024. Available from: https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-zolbetuximab-clzb-chemotherapy-gastric-or-gastroesophageal-junction-adenocarcinoma.
- Arias-Pinilla GA, Dalgleish AG, Mudan S, Bagwan I, Walker AJ, Modjtahedi H. Development of novel monoclonal antibodies against CD109 overexpressed in human pancreatic cancer. Oncotarget. 2018;9(28):19994-20007.
doi pubmed - Dinse GE, Lagakos SW. Nonparametric estimation of lifetime and disease onset distributions from incomplete observations. Biometrics. 1982;38(4):921-932.
pubmed - Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, Jemal A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74(3):229-263.
doi pubmed - Institute NC. SEER*Explorer: An interactive website for SEER cancer statistics [Internet]. Surveillance Research Program, National Cancer Institute. 2024. Available from: https://seer.cancer.gov/statistics-network/explorer/application.html?site=18&data_type=4&graph_type=5&compareBy=stage&chk_stage_104=104&chk_stage_105=105&chk_stage_106=106&chk_stage_107=107&series=9&sex=1&race=1&age_range=1&advopt_precision=1&advopt_show_ci=on&hdn_view=1#resultsRegion1.
- Atmaca A, Werner D, Pauligk C, Steinmetz K, Wirtz R, Altmannsberger HM, Jager E, et al. The prognostic impact of epidermal growth factor receptor in patients with metastatic gastric cancer. BMC Cancer. 2012;12:524.
doi pubmed - Aldape KD, Ballman K, Furth A, Buckner JC, Giannini C, Burger PC, Scheithauer BW, et al. Immunohistochemical detection of EGFRvIII in high malignancy grade astrocytomas and evaluation of prognostic significance. J Neuropathol Exp Neurol. 2004;63(7):700-707.
doi pubmed - Gan HK, Cvrljevic AN, Johns TG. The epidermal growth factor receptor variant III (EGFRvIII): where wild things are altered. FEBS J. 2013;280(21):5350-5370.
doi pubmed - Fuse N, Kuboki Y, Kuwata T, Nishina T, Kadowaki S, Shinozaki E, Machida N, et al. Prognostic impact of HER2, EGFR, and c-MET status on overall survival of advanced gastric cancer patients. Gastric Cancer. 2016;19(1):183-191.
doi pubmed - Chen Y, Guo SY, Guo W. The association between EGFR expression and clinical pathology characteristics in gastric cancer. Open Life Sciences. 2016;11(1):318-321.
- Ueda S, Ogata S, Tsuda H, Kawarabayashi N, Kimura M, Sugiura Y, Tamai S, et al. The correlation between cytoplasmic overexpression of epidermal growth factor receptor and tumor aggressiveness: poor prognosis in patients with pancreatic ductal adenocarcinoma. Pancreas. 2004;29(1):e1-8.
doi pubmed - Kappler M, Dauter K, Reich W, Bethmann D, Schwabe M, Rot S, Wickenhauser C, et al. Prognostic impact of cytoplasmatic EGFR upregulation in patients with oral squamous cell carcinoma: A pilot study. Mol Clin Oncol. 2020;13(6):88.
doi pubmed - Tang D, Liu CY, Shen D, Fan S, Su X, Ye P, Gavine PR, et al. Assessment and prognostic analysis of EGFR, HER2, and HER3 protein expression in surgically resected gastric adenocarcinomas. Onco Targets Ther. 2015;8:7-14.
doi pubmed - Nagatsuma AK, Aizawa M, Kuwata T, Doi T, Ohtsu A, Fujii H, Ochiai A. Expression profiles of HER2, EGFR, MET and FGFR2 in a large cohort of patients with gastric adenocarcinoma. Gastric Cancer. 2015;18(2):227-238.
doi pubmed - Wang D, Wang B, Wang R, Zhang Z, Lin Y, Huang G, Lin S, et al. High expression of EGFR predicts poor survival in patients with resected T3 stage gastric adenocarcinoma and promotes cancer cell survival. Oncol Lett. 2017;13(5):3003-3013.
doi pubmed - Inokuchi M, Murayama T, Hayashi M, Takagi Y, Kato K, Enjoji M, Kojima K, et al. Prognostic value of co-expression of STAT3, mTOR and EGFR in gastric cancer. Exp Ther Med. 2011;2(2):251-256.
doi pubmed - Pinto C, Di Fabio F, Barone C, Siena S, Falcone A, Cascinu S, Rojas Llimpe FL, et al. Phase II study of cetuximab in combination with cisplatin and docetaxel in patients with untreated advanced gastric or gastro-oesophageal junction adenocarcinoma (DOCETUX study). Br J Cancer. 2009;101(8):1261-1268.
doi pubmed - Waddell T, Chau I, Cunningham D, Gonzalez D, Okines AF, Okines C, Wotherspoon A, et al. Epirubicin, oxaliplatin, and capecitabine with or without panitumumab for patients with previously untreated advanced oesophagogastric cancer (REAL3): a randomised, open-label phase 3 trial. Lancet Oncol. 2013;14(6):481-489.
doi pubmed - Al-Janaby T, Nahi N, Seddon A, Bagwan I, Khelwatty S, Modjtahedi H. The combination of afatinib with dasatinib or miransertib results in synergistic growth inhibition of stomach cancer cells. World J Oncol. 2024;15(2):192-208.
doi pubmed - Okamoto I, Kenyon LC, Emlet DR, Mori T, Sasaki J, Hirosako S, Ichikawa Y, et al. Expression of constitutively activated EGFRvIII in non-small cell lung cancer. Cancer Sci. 2003;94(1):50-56.
doi pubmed - Montano N, Cenci T, Martini M, D'Alessandris QG, Pelacchi F, Ricci-Vitiani L, Maira G, et al. Expression of EGFRvIII in glioblastoma: prognostic significance revisited. Neoplasia. 2011;13(12):1113-1121.
doi pubmed - Wheeler SE, Egloff AM, Wang L, James CD, Hammerman PS, Grandis JR. Challenges in EGFRvIII detection in head and neck squamous cell carcinoma. PLoS One. 2015;10(2):e0117781.
doi pubmed - Sherif O, Khelwatty SA, Bagwan I, Seddon AM, Dalgleish A, Mudan S, Modjtahedi H. Expression of EGFRvIII and its co-expression with wild-type EGFR, or putative cancer stem cell biomarkers CD44 or EpCAM are associated with poorer prognosis in patients with hepatocellular carcinoma. Oncol Rep. 2024;52(6).
doi pubmed - Viana-Pereira M, Lopes JM, Little S, Milanezi F, Basto D, Pardal F, Jones C, et al. Analysis of EGFR overexpression, EGFR gene amplification and the EGFRvIII mutation in Portuguese high-grade gliomas. Anticancer Res. 2008;28(2A):913-920.
pubmed - Iurlaro R, Waldhauer I, Planas-Rigol E, Bonfill-Teixidor E, Arias A, Nicolini V, Freimoser-Grundschober A, et al. A novel EGFRvIII T-cell bispecific antibody for the treatment of glioblastoma. Mol Cancer Ther. 2022;21(10):1499-1509.
doi pubmed - Ghaderi A, Vasei M, Maleck-Hosseini SA, Gharesi-Fard B, Khodami M, Doroudchi M, Modjtahedi H. The expression of c-erbB-1 and c-erbB-2 in Iranian patients with gastric carcinoma. Pathol Oncol Res. 2002;8(4):252-256.
doi pubmed - Tewari M, Kumar A, Mishra RR, Kumar M, Shukla HS. HER2 expression in gastric and gastroesophageal cancer: report from a tertiary care hospital in North India. Indian J Surg. 2015;77(Suppl 2):447-451.
doi pubmed - Ugras N, Ozgun G, Ocakoglu G, Yerci O, Ozturk E. Relationship between HER-2, COX-2, p53 and clinicopathologic features in gastric adenocarcinoma. Do these biomarkers have any prognostic significance? Turk J Gastroenterol. 2014;25(Suppl 1):176-181.
doi pubmed - Wang S, Zheng G, Chen L, Xiong B. Effect of HER-2/neu over-expression on prognosis in gastric cancer: a meta-analysis. Asian Pac J Cancer Prev. 2011;12(6):1417-1423.
pubmed - Janjigian YY, Werner D, Pauligk C, Steinmetz K, Kelsen DP, Jager E, Altmannsberger HM, et al. Prognosis of metastatic gastric and gastroesophageal junction cancer by HER2 status: a European and USA International collaborative analysis. Ann Oncol. 2012;23(10):2656-2662.
doi pubmed - He C, Bian XY, Ni XZ, Shen DP, Shen YY, Liu H, Shen ZY, et al. Correlation of human epidermal growth factor receptor 2 expression with clinicopathological characteristics and prognosis in gastric cancer. World J Gastroenterol. 2013;19(14):2171-2178.
doi pubmed - Shitara K, Bang YJ, Iwasa S, Sugimoto N, Ryu MH, Sakai D, Chung HC, et al. Trastuzumab Deruxtecan in previously treated HER2-positive gastric cancer. N Engl J Med. 2020;382(25):2419-2430.
doi pubmed - Palyi-Krekk Z, Barok M, Kovacs T, Saya H, Nagano O, Szollosi J, Nagy P. EGFR and ErbB2 are functionally coupled to CD44 and regulate shedding, internalization and motogenic effect of CD44. Cancer Lett. 2008;263(2):231-242.
doi pubmed - Bao W, Fu HJ, Xie QS, Wang L, Zhang R, Guo ZY, Zhao J, et al. HER2 interacts with CD44 to up-regulate CXCR4 via epigenetic silencing of microRNA-139 in gastric cancer cells. Gastroenterology. 2011;141(6):2076-2087.e6.
doi pubmed - Perez A, Neskey DM, Wen J, Pereira L, Reategui EP, Goodwin WJ, Carraway KL, et al. CD44 interacts with EGFR and promotes head and neck squamous cell carcinoma initiation and progression. Oral Oncol. 2013;49(4):306-313.
doi pubmed - Yu H, Gong X, Luo X, Han W, Hong G, Singh B, Tang CK. Co-expression of EGFRvIII with ErbB-2 enhances tumorigenesis: EGFRvIII mediated constitutively activated and sustained signaling pathways, whereas EGF-induced a transient effect on EGFR-mediated signaling pathways. Cancer Biol Ther. 2008;7(11):1818-1828.
doi pubmed - Schuler MH, Al-Batran S-E, Zvirbule Z, Manikhas G, Lordick F, Rusyn AV, et al. Expression of Claudin 18.2 and HER2 in gastric, gastroesophageal junction, and esophageal cancers: Results from the FAST study. Journal of Clinical Oncology. 2017;35(15_suppl):4038.
- Moran D, Maurus D, Rohde C, Arozullah A. Prevalence of CLDN18.2, HER2 and PD-L1 in gastric cancer samples. Annals of Oncology. 2018;29:viii32.
- Sheng T, Sundar R, Srivastava S, Ong X, Tay ST, Ma H, et al. Spatial profiling of Patient-Matched HER2 positive gastric cancer reveals resistance mechanisms to Trastuzumab and Trastuzumab Deruxtecan. medRxiv. 2024:2024.
- Adachi K, Sakurai Y, Ichinoe M, Tadehara M, Tamaki A, Kesen Y, Kato T, et al. CD109 expression in tumor cells and stroma correlates with progression and prognosis in pancreatic cancer. Virchows Arch. 2022;480(4):819-829.
doi pubmed - Lee KY, Shueng PW, Chou CM, Lin BX, Lin MH, Kuo DY, Tsai IL, et al. Elevation of CD109 promotes metastasis and drug resistance in lung cancer via activation of EGFR-AKT-mTOR signaling. Cancer Sci. 2020;111(5):1652-1662.
doi pubmed - Arias-Pinilla GA, Modjtahedi H. Therapeutic application of monoclonal antibodies in pancreatic cancer: advances, challenges and future opportunities. Cancers (Basel). 2021;13(8).
doi pubmed - Kodama H, Murata S, Ishida M, Yamamoto H, Yamaguchi T, Kaida S, Miyake T, et al. Prognostic impact of CD44-positive cancer stem-like cells at the invasive front of gastric cancer. Br J Cancer. 2017;116(2):186-194.
doi pubmed - Khelwatty SA, Essapen S, Bagwan I, Green M, Seddon AM, Modjtahedi H. Co-expression and prognostic significance of putative CSC markers CD44, CD133, wild-type EGFR and EGFRvIII in metastatic colorectal cancer. Oncotarget. 2019;10(18):1704-1715.
doi pubmed - Cao W, Xing H, Li Y, Tian W, Song Y, Jiang Z, Yu J. Claudin18.2 is a novel molecular biomarker for tumor-targeted immunotherapy. Biomark Res. 2022;10(1):38.
doi pubmed - Jun KH, Kim JH, Jung JH, Choi HJ, Chin HM. Expression of claudin-7 and loss of claudin-18 correlate with poor prognosis in gastric cancer. Int J Surg. 2014;12(2):156-162.
doi pubmed - Kim M, Kang BW, Park J, Baek JH, Kim JG. Expression of claudin 18.2 in poorly cohesive carcinoma and its association with clinicopathologic parameters in East Asian patients. Pathol Res Pract. 2024;263:155628.
doi pubmed - Kwak Y, Kim TY, Nam SK, Hwang HJ, Han D, Oh HJ, Kong SH, et al. Clinicopathologic and molecular characterization of stages II-IV gastric cancer with Claudin 18.2 expression. Oncologist. 2025;30(2).
doi pubmed - Yan P, Dong Y, Zhang F, Zhen T, Liang J, Shi H, Han A. Claudin18.2 expression and its clinicopathological feature in adenocarcinoma from various parts. J Clin Pathol. 2024.
doi pubmed - Wang C, Wang Y, Chen J, Wang Y, Pang C, Liang C, Yuan L, et al. CLDN18.2 expression and its impact on prognosis and the immune microenvironment in gastric cancer. BMC Gastroenterol. 2023;23(1):283.
doi pubmed - Moraes FCA, Rodrigues Sobreira LE, Cavalcanti Souza ME, Burbano RMR. The role of CLDN18.2 in gastric cancer prognosis: a systematic review and meta-analysis. Biomarkers. 2024;29(8):528-538.
doi pubmed - Kubota Y, Kawazoe A, Mishima S, Nakamura Y, Kotani D, Kuboki Y, Bando H, et al. Comprehensive clinical and molecular characterization of claudin 18.2 expression in advanced gastric or gastroesophageal junction cancer. ESMO Open. 2023;8(1):100762.
doi pubmed - Arnold A, Daum S, von Winterfeld M, Berg E, Hummel M, Rau B, Stein U, et al. Prognostic impact of Claudin 18.2 in gastric and esophageal adenocarcinomas. Clin Transl Oncol. 2020;22(12):2357-2363.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
World Journal of Oncology is published by Elmer Press Inc.