| World Journal of Oncology, ISSN 1920-4531 print, 1920-454X online, Open Access |
| Article copyright, the authors; Journal compilation copyright, World J Oncol and Elmer Press Inc |
| Journal website https://wjon.elmerpub.com |
Review
Volume 16, Number 2, April 2025, pages 143-151
Tumor Necrosis Factor-Alpha and Its Association With Breast Cancer: A Systematic Review
Nur Qodira, b, Didit Pramudhitoa, Legiranc, d, Zen Hafyc, Muhammad Baharul Imanb, Fara Syafirab, Putri Mahirah Afladhantib, Raehan Satya Daenasab, Bima Indrab
aDepartment of Surgery, University of Sriwijaya/Mohammad Hoesin General Hospital,
Palembang, Indonesia
bFaculty of Medicine, University of Sriwijaya, Palembang,
Indonesia
cDepartment of Biomedicine, University of Sriwijaya, Palembang,
Indonesia
dCorresponding Author: Legiran, Department of Biomedicine, University
of Sriwijaya, Palembang 30139, Indonesia
Manuscript submitted January 12, 2025, accepted March 18, 2025, published online March 22,
2025
Short title: TNF-α and Breast Cancer
doi: https://doi.org/10.14740/wjon2532
| Abstract | ▴Top |
Background: Tumor necrosis factor-alpha (TNF-α) is a pro-inflammatory cytokine implicated in the pathogenesis and progression of various cancers, including breast cancer. Elevated TNF-α levels have been associated with cancer progression, metastasis, and treatment outcomes. This systematic review aimed to synthesize existing evidence on the relationship between TNF-α levels and breast cancer.
Methods: A systematic search of observational studies published from inception to June 2024 was conducted in PubMed, ScienceDirect, Sage Journals, and Google Scholar to identify studies examining TNF-α levels in breast cancer patients compared to healthy controls, as well as its association with metastasis, response to chemotherapy, and survival outcomes. Inclusion criteria were applied to select eligible studies, resulting in nine studies that met the criteria for this review.
Results: Eight eligible studies reported that breast cancer patients exhibited higher TNF-α levels than healthy controls. Two studies indicated that TNF-α levels were elevated in patients with metastatic breast cancer. Additionally, two studies found that patients with higher TNF-α levels tended to have a poorer response to chemotherapy. One study revealed that patients with elevated TNF-α levels had a lower mean survival time.
Conclusions: Elevated TNF-α levels are significantly associated with breast cancer progression, metastasis, and poorer treatment outcomes. These findings underscore the potential of TNF-α as a biomarker for breast cancer prognosis and therapeutic response. Further research is warranted to explore the underlying mechanisms and validate TNF-α as a target for therapeutic intervention in breast cancer management.
Keywords: Breast cancer; Tumor necrosis factor-alpha; Biomarker
| Introduction | ▴Top |
Inflammation plays a crucial role in cancer development and progression by inducing genetic and epigenetic changes that alter cellular pathways responsible for maintaining normal homeostasis [1]. Various inflammatory mediators such as cytokines, free radicals, and growth factors contribute to genetic mutations in tumor suppressor genes, DNA methylation, and post-translational modifications, ultimately fueling tumor progression [1]. Chronic inflammation has been recognized as a hallmark of cancer, influencing tumor response to treatment and promoting tumor progression in various cancers [2]. Even in cancers without preceding inflammation, tumor-elicited inflammation, inflammatory secretions, and infiltrating immune cells play critical roles in cancer initiation, promotion, and progression to metastasis [3]. The inflammatory microenvironment, comprising inflammatory cells and signaling molecules, is essential for the malignant progression of transformed cells [4]. Inflammatory responses affect cancer development, including DNA replication, tumor cell survival, angiogenesis, metastasis, and immune evasion [5].
In breast cancer, inflammation significantly influences the disease’s development and progression. Inflammatory processes have been linked to various aspects of breast cancer, including migration, invasion, metastasis, and treatment response [6]. The inflammatory microenvironment in breast cancer, characterized by inflammatory cytokines and immune cells, can promote tumor growth and metastasis [7]. Infiltration of macrophages producing cytokines into the tumor microenvironment is associated with increased angiogenesis, metastasis, and decreased survival in breast cancer patients [8]. Molecular mechanisms, such as the tumor necrosis factor-alpha (TNF-α)-IKK-YAP/p65-HK2 signaling axis, drive inflammation-driven migration in breast cancer cells, linking inflammation to metastasis [9]. The upregulation of sialylated N-glycans and integrins is associated with breast cancer brain metastasis, emphasizing the role of glycomics in metastatic spread [10]. Additionally, inflammation and specific signaling pathways, such as the A20/TNFAIP3-CDC20-CASP1 axis, promote inflammation-mediated metastatic disease in triple-negative breast cancer [11]. The chemokine (CC motif) ligand (CCL)28 activates the mitogen-activated protein kinases (MAPK) pathway, leading to increased proliferation and metastasis of breast cancer cells [12].
Tumor necrosis factor-alpha (TNF-α) is a well-documented pro-inflammatory cytokine upregulated in breast cancer. High levels of TNF-α have been associated with breast cancer recurrence [13]. In breast cancer, TNF-α influences the inflammatory profile of stromal cells, such as cancer-associated fibroblasts (CAFs) and mesenchymal stem cells (MSCs), by promoting the expression of inflammatory chemokines like CCL2, C-X-C motif chemokine ligand 8 (CXCL8), and CCL5, which are pro-tumorigenic [14]. TNF-α is linked to lymph node involvement and enhances tumor cell metastasis, promoting cancer progression [15]. TNF-α increases breast cancer stem-like cells by upregulating TAZ expression via the non-canonical nuclear factor kappa B (NF-κB) pathway, maintaining a stem cell-like phenotype [13]. In breast cancer cells, TNF-α confers resistance to apoptosis by activating the NF-κB pathway and upregulating antiapoptotic proteins like B-cell lymphoma 2 (Bcl-2), promoting cell survival and tumorigenic potential [16]. TNF-α’s pro-inflammatory and pro-tumorigenic effects in breast cancer underscore its significance in driving cancer progression and influencing the tumor microenvironment.
The role of TNF-α in breast cancer spans various stages of tumorigenesis, including cellular transformation, promotion, survival, proliferation, invasion, angiogenesis, and metastasis [17]. Studies have linked TNF-α to lymph node involvement and enhanced tumor cell metastasis in breast cancer, highlighting its pro-tumorigenic properties [15]. TNF-α promotes tumor growth through a positive feedback loop involving TNFR1/NF-κB/p-STAT3/HBXIP/TNFR1, shedding light on its growth-promoting mechanisms in breast cancer [18]. TNF-α’s role in breast cancer progression, metastasis, and treatment response is mediated by its effects on stem cell-like properties, apoptosis resistance, gene regulation, adhesion molecules, inflammation, and metastasis [19, 20].
Notwithstanding significant strides in breast cancer investigation and care, the disease remains a chief global health problem with high morbidity and mortality. In cancer development and progression, inflammation has a major role to play among the inflammatory cytokines; TNF-α has drawn much attention due to its potential implications in cancer biology. TNF-α is known to affect tumor growth, metastases, as well as the immune response against cancers, making it an interesting biomarker for understanding breast cancer dynamics. Nevertheless, the existing literature on TNF-α and breast cancer varies widely and at times displays contradictions; this makes it necessary to comprehensively synthesize all evidence available. This review aims to clarify the relationship between TNF-α levels and breast cancer by systematically reviewing the available studies, thereby providing insights that could improve diagnostic and prognostic techniques, consequently guiding therapeutic interventions.
| Materials and Methods | ▴Top |
A systematic search was performed to investigate the association between TNF-α levels and breast cancer, adhering to the guidelines outlined in the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 framework. The search strategy targeted observational studies published in reputable databases, including PubMed, ScienceDirect, Sage Journals, and Google Scholar, up to June 2024. A combination of keywords and medical subject headings (MeSH) terms such as “tumor necrosis factor alpha,” “TNF-α,” “breast cancer,” “breast carcinoma,” “metastasis,” “chemotherapy response,” and “survival” was utilized, with Boolean operators (AND, OR) applied to refine the results. Studies were included if they were observational, published in English or Indonesian, and directly examined the relationship between TNF-α and breast cancer. To ensure comparability, included studies needed to have control groups matched for key demographic factors, particularly age. Studies were excluded if they involved non-human subjects, lacked relevant data on TNF-α levels in breast cancer patients, or did not provide adequate information on control group characteristics to minimize selection bias (Table 1).
 Click to view |
Table 1. Eligibility Criteria for This
Review |
Reviewers independently screened titles and abstracts to identify eligible studies, and discussions resolved disagreements. The selected studies were organized using the Mendeley Reference Manager to ensure efficient data handling. Extracted information included the study title, author details, demographics (such as age, sex, and study location), methodological approaches, and key findings. If necessary, the authors of original publications were contacted to provide additional data or clarify unclear points.
To assess the quality and reliability of the included studies, the Revised Risk of Bias Assessment Tool for Nonrandomized Studies of Interventions (RoBANS 2) was employed. RoBANS 2 is an updated framework designed to evaluate the risk of bias in nonrandomized studies, such as cohort, case-control, and cross-sectional studies. It refines the original RoBANS tool by incorporating additional bias domains and improving assessment reliability. Each reviewer independently evaluated the studies for potential bias, and any discrepancies in assessments were resolved collectively. Data from the selected studies were analyzed to examine the relationship between TNF-α and breast cancer, focusing on correlation coefficients, statistical significance, and P values. The findings were then synthesized qualitatively to identify patterns and trends across studies, offering a comprehensive understanding of how TNF-α contributes to breast cancer progression, metastasis, and treatment outcomes. This systematic approach ensures the robustness of the analysis and provides a valuable foundation for future research on the role of TNF-α in breast cancer.
| Results | ▴Top |
A search was conducted across multiple electronic databases, yielding many articles. For instance, an initial search on PubMed identified 865 articles. To refine the results, a filter for observational studies was applied, reducing the number of articles to 37. These articles were then meticulously reviewed for relevance to the research focus and evaluated based on the predetermined inclusion and exclusion criteria. From the database searches, 15 articles were deemed potentially eligible based on their titles. After duplicate records were removed and full-text reviews were performed, studies that did not report outcomes related to breast cancer and TNF-α were excluded [21, 22]. After all, nine articles fulfilled all inclusion criteria and were included in the systematic review. This thorough screening and selection process is summarized in the final study flow diagram (Fig. 1).
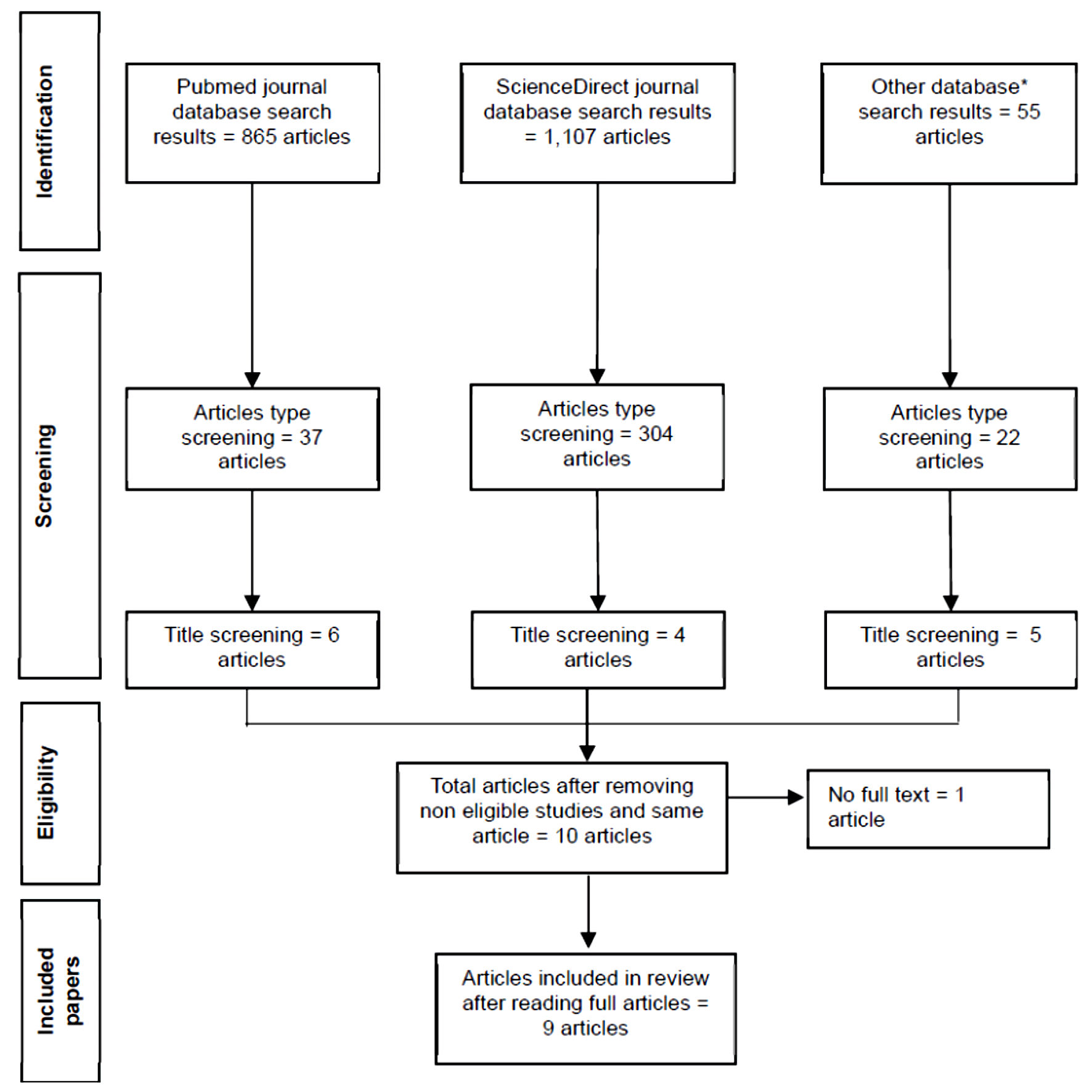 Click for large image |
Figure 1. PRISMA flowchart. PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses. |
A total of nine studies met the eligibility criteria for this systematic review, involving a combined sample of 1,607 women, of which 918 were breast cancer patients and 689 were healthy controls. The studies were geographically diverse, with two conducted in Indonesia [23, 24] and one each in China [25], France [26], Turkey [27], Greece [28], Saudi Arabia [29], Iran [30], and Iraq [31]. Seven studies employed a case-control design [24-29, 31], while two studies used a cross-sectional design [23, 30]. The mean age of participants ranged from 37.4 years to 61.3 years in the breast cancer group and 34.7 years to 57.2 years in the healthy control group, with the youngest participant being 20 years old and the oldest being 80 years old. Regarding demographic characteristics, five studies reported that the breast cancer group had a slightly higher mean age than the healthy control group, while one study found that the control group had a marginally higher mean age. Two studies provided statistical analyses indicating no significant differences in mean age between the groups with respect to TNF-α levels. The remaining two studies did not report specific age data; however, their analyses confirmed that there were no significant differences in age between breast cancer patients and healthy controls. It is noteworthy that one study did not include a healthy control group.
The risk of bias assessment using the RoBANS 2 indicated that three studies had a high risk of bias [24, 30, 31], three had some concerns [23, 25, 27], and three had a low risk of bias [26, 28, 29] (Table 2).
 Click to view |
Table 2. Risk of Bias Assessment Using Revised
Risk of Bias Assessment Tool for Nonrandomized Studies of Interventions (RoBANS
2) |
A closer examination of each study reveals a consistent trend in the association between TNF-α levels and breast cancer characteristics. In the study by Moaiedi et al, a cross-sectional analysis of 50 subjects showed that TNF-α mRNA expression was higher in patients without surgery compared to controls, although this increase was not statistically significant [30]. In contrast, Gharib et al demonstrated in a case-control study of 200 participants that breast cancer patients had significantly elevated serum TNF-α levels compared to healthy women [29]. Similarly, Papadopoulou et al reported markedly higher TNF-α serum levels in breast cancer patients relative to controls and further linked high TNF-α levels with larger tumor size, poor differentiation, increased lymph node involvement, and shorter mean survival [28]. Berberoglu et al observed that patients exhibited significantly higher TNF-α levels than controls, and noted a significant reduction in TNF-α following chemotherapy, which correlated with clinical response [27]. A study from Indonesia found a significant negative correlation between TNF-α levels and clinical response in 38 patients undergoing anthracycline-based neoadjuvant chemotherapy, suggesting that elevated TNF-α may predict a poorer response to treatment [23]. Another study from Indonesia reported a statistically significant association between serum TNF-α levels and metastases in a cohort of 50 patients [24]. In a large-scale case-control study conducted in France, involving 453 breast cancer patients and 453 controls, higher TNF-α levels were significantly associated with an increased risk of breast cancer after adjustment for confounders [26]. Study in China demonstrated that patients with stage III breast cancer had significantly higher serum TNF-α levels compared to healthy controls, with TNF-α correlating significantly with clinical tumor stage and lymph node metastasis [25]. Lastly, Mohammed in Iraq observed that while TNF-α levels were significantly elevated in patients with ductal carcinoma and those with lymph node metastasis, there was no significant difference in TNF-α levels between metastatic and non-metastatic groups in a study of 90 participants [31].
Overall, the results consistently demonstrated that breast cancer patients had higher TNF-α levels than healthy controls, as reported in eight studies [24-31]. Additionally, two studies highlighted that TNF-α levels were particularly elevated in patients with metastatic breast cancer [25, 28], and one study did not find a significant association between TNF-α levels and the presence of metastasis [31]. Furthermore, two studies identified a correlation between high TNF-α levels and a negative response to chemotherapy [23, 27]. Lastly, one study found that patients with elevated TNF-α levels had a lower mean survival time. A summary of the included studies is shown in Table 3 [23-31].
 Click to view |
Table 3. Summary of Included
Studies |
| Discussion | ▴Top |
The strength of the positive association between TNF-α levels and breast cancer incidence appears well supported, as three of the supporting studies had a low risk of bias, two had some concerns, and only two were rated as high risk. The main sources of bias in the high-risk studies were related to comparability of the target group and target group selection, which could affect the validity of the findings but are unlikely to overturn the overall trend observed. However, the association between TNF-α levels and metastasis remains uncertain. While two studies reported a positive association, both had some concerns or a high risk of bias, particularly in participant selection and confounder control. Additionally, the one study that did not find an association also had a high risk of bias. The lack of correlation between TNF-α levels and metastasis in the Iraq study may indicate that other factors influence chemotherapy response in that population. Chemotherapy response in breast cancer patients is affected by multiple factors, including the tumor microenvironment, hormone receptor status, and proliferation indices [32, 33]. Additionally, levels of other cytokines, such as interleukin (IL)-6, have been associated with poor chemotherapy response, suggesting that TNF-α alone may not fully predict treatment outcomes [34]. Geographic factors, such as healthcare accessibility, also play a role, as disparities in treatment availability and compliance can impact chemotherapy effectiveness [35]. These variables highlight the complexity of chemotherapy response and the need for further research to clarify TNF-α’s role in different populations. Given these limitations, the interpretation of TNF-α as a marker for metastasis remains inconclusive, and further studies with stronger methodological designs are needed to clarify this relationship. Regarding TNF-α levels and chemotherapy response, although the two supporting studies did not have opposing findings, both had some concerns about confounder control, making it unclear whether other factors influenced their results. Despite this, the consistency of the findings suggests that higher TNF-α levels may be associated with poorer chemotherapy response, though further research with better bias minimization is necessary to strengthen this conclusion.
TNF-α significantly impacts breast cancer progression and treatment outcomes through various biological mechanisms, including promoting tumor growth, metastasis, inflammation, and resistance to chemotherapy. Understanding these mechanisms is crucial for developing targeted therapeutic strategies for breast cancer patients. This review highlighted several key findings regarding TNF-α’s role in breast cancer. TNF-α substantially influences breast cancer by interacting with the TNF-TNFR2 axis, which directly affects cancer cell behavior by promoting tumorigenesis, invasion, and metastasis [1]. This cytokine alters the tumor microenvironment, impacting disease progression and treatment outcomes [2]. A critical mechanism by which TNF-α affects breast cancer is through the upregulation of TAZ expression via the non-canonical NF-κB pathway, increasing the population of breast cancer stem-like cells. These cells are known for their role in tumor initiation, progression, and resistance to therapy [3]. Furthermore, TNF-α is linked to genetic variations that may elevate breast cancer risk, emphasizing its role in the early stages of cancer development [4]. It also induces changes in gene expression related to metastasis, which can significantly impact treatment efficacy [5]. The involvement of TNF-α in a positive feedback loop involving TNFR1/NF-κB/p-STAT3/HBXIP/TNFR1 further highlights its role in sustaining tumor growth and influencing treatment response [36]. Additionally, TNF-α regulates adhesion molecules, contributing to cancer cell dissemination and metastasis, which are critical factors in treatment outcomes [37].
The overexpression of metalloproteinases (MMPs) induced by TNF-α is another significant pathway driving cancer progression and metastasis. MMPs facilitate the breakdown of extracellular matrix components, promoting tumor invasion and metastasis [38]. These diverse biological mechanisms underscore the complexity of TNF-α’s role in breast cancer, influencing various stages of tumor development and response to treatment. TNF-α promotes breast cancer progression by enhancing tumor growth, migration, and survival of malignant cells while suppressing adaptive immune responses and contributing to resistance to hormonal and chemotherapeutic agents [15]. This review results suggest that TNF-α’s ability to increase breast cancer stem-like cells via the non-canonical NF-κB pathway plays a pivotal role in maintaining a population of cells capable of driving tumor recurrence and resistance to therapy [13]. Moreover, TNF-α confers resistance to apoptosis in breast cancer cells by activating the NF-κB pathway and upregulating antiapoptotic proteins such as Bcl-2, promoting cell survival and enhancing the tumorigenic potential of breast cancer cells [16]. The induction of MMPs and activation of the ATM pathway by TNF-α further facilitate tumor metastasis, highlighting its role in promoting cancer cell invasion and dissemination [39]. TNF-α’s role in promoting resistance to chemotherapy is significant, as it modulates the expression of genes and proteins involved in treatment resistance. By activating the NF-κB pathway, TNF-α increases the expression of inhibitors of apoptosis proteins (IAPs) and other factors that contribute to treatment resistance [40]. This cytokine also influences the inflammatory profile of the tumor microenvironment, which can lead to resistance to chemotherapeutic agents [41]. This review also highlighted that TNF-α’s ability to upregulate TAZ expression and maintain a stem-cell-like phenotype in breast cancer cells contributes to resistance against conventional therapies, as these stem-like cells are often less responsive to treatment. This mechanism aligns with the findings from two studies in this review, which reported that higher TNF-α levels were associated with poor chemotherapy response [13]. The resistance may be explained by the role of TNF-α in promoting cancer stem-like properties, which have been linked to enhanced survival pathways, reduced apoptosis, and increased drug resistance mechanisms [42, 43]. These findings suggest that TNF-α not only plays a role in breast cancer progression but may also serve as a potential target to improve chemotherapy efficacy. Understanding these mechanisms is crucial for developing effective therapeutic strategies that can overcome TNF-α-mediated resistance and improve treatment outcomes for breast cancer patients.
The systematic review has important implications for clinical practice and research in breast cancer. Hence, the review confirms that breast cancer patients have elevated TNF-α levels compared to those without cancer, based on results from different geographical areas and rigorous methodology assessments. In addition, there were similar findings across all studies that support the role for TNF-α as a potential biomarker for the diagnosis and monitoring of breast cancers. The fact that TNF-α is associated with metastasis, chemotherapy response, and survival outcomes also suggests its potential to predict disease aggressiveness or treatment efficiency. The availability of this information enables clinicians to utilize levels of TNF-α as an ancillary diagnostic tool as well as a prognostic marker, with the possibility to change treatment decisions affecting patient prognosis.
Nonetheless, some limitations need to be considered. First, the studies included in this systematic review were retrospective and thus cannot establish evidence for causality between high levels of TNF-a and breast cancer outcomes. Nonetheless, utilizing RoBANS 2 ensures dependability regarding the included studies. Second, there was enormous variation in study design, populations recruited, and even methods used in measuring TNF-α, leading to significant heterogeneity among the studies that may have been introduced. Another limitation of this review is the absence of studies from Western countries. This may be due to the tendency of research in those regions to focus on large-scale cytokine screening rather than individual cytokines like TNF-α. As a result, relevant data from Western populations may not have been captured within our search criteria. This limits the generalizability of our findings, as differences in genetic, environmental, and lifestyle factors between populations could influence TNF-α levels and their association with breast cancer.
| Conclusions | ▴Top |
The findings of this review indicate that TNF-α levels tend to be higher in breast cancer patients compared to healthy controls. Elevated TNF-α levels have also been observed in patients with metastatic breast cancer, though some findings suggest no significant association. Additionally, higher TNF-α levels have been linked to poor chemotherapy response and reduced survival time. These results suggest a potential role of TNF-α in breast cancer progression and treatment outcomes.
Acknowledgments
We sincerely thank all individuals and institutions who supported this review, including those who provided access to resources and constructive feedback. We also appreciate the researchers whose work contributed to this study.
Financial Disclosure
This study does not receive any funding.
Conflict of Interest
The authors declare no conflict of interest regarding this review.
Author Contributions
Conceptualization: NQ, LG, ZH, DP, and BI. Methodology: NQ, ZH, MBI, and BI. Data analysis: BI, MBI, FS, and PMA. Resources: BI, MBI, FS, RSD, and PMA. Writing - original draft preparation: BI, FS, PMA, RSD, and MBI. Writing - review and editing: NQ, BI, ZH, and LG. Visualization: BI, FS, PMA, RSD, and MBI. Supervision: NQ, ZH, DP, and LG. Administration: BI, FS, PMA, RSD, and MBI. All authors have read and agreed to the published version of the manuscript.
Data Availability
The authors declare that data supporting the findings of this study are available within the article.
| References | ▴Top |
- Hussain SP, Harris CC. Inflammation and cancer: an ancient
link with novel potentials. Int J Cancer. 2007;121(11):2373-2380.
doi pubmed - Lavy M, Gauttier V, Poirier N, Barille-Nion S, Blanquart C.
Specialized pro-resolving mediators mitigate cancer-related inflammation: role of
tumor-associated macrophages and therapeutic opportunities. Front Immunol.
2021;12:702785.
doi pubmed - Shadhu K, Xi C. Inflammation and pancreatic cancer: an
updated review. Saudi J Gastroenterol. 2019;25(1):3-13.
doi pubmed - Lu H, Ouyang W, Huang C. Inflammation, a key event in cancer
development. Mol Cancer Res. 2006;4(4):221-233.
doi pubmed - Yu X, Lian B, Wang L, Zhang Y, Dai E, Meng F, Liu D, et al.
The pan-cancer analysis of gene expression patterns in the context of inflammation. Mol Biosyst.
2014;10(9):2270-2276.
doi pubmed - Zhao Y, Kong X, Li X, Yan S, Yuan C, Hu W, Yang Q.
Metadherin mediates lipopolysaccharide-induced migration and invasion of breast cancer cells.
PLoS One. 2011;6(12):e29363.
doi pubmed - Nagahashi M, Yamada A, Katsuta E, Aoyagi T, Huang WC,
Terracina KP, Hait NC, et al. Targeting the SphK1/S1P/S1PR1 axis that links obesity, chronic
inflammation, and breast cancer metastasis. Cancer Res. 2018;78(7):1713-1725.
doi pubmed - Gao Y, Yang Y, Yuan F, Huang J, Xu W, Mao B, Yuan Z, et al.
TNFalpha-YAP/p65-HK2 axis mediates breast cancer cell migration. Oncogenesis.
2017;6(9):e383.
doi pubmed - Yu L, Maishi N, Akahori E, Hasebe A, Takeda R, Matsuda AY,
Hida Y, et al. The oral bacterium Streptococcus mutans promotes tumor metastasis by inducing
vascular inflammation. Cancer Sci. 2022;113(11):3980-3994.
doi pubmed - Kelly LM, Buggy Y, Hill A, O'Donovan N, Duggan C, McDermott
EW, O'Higgins NJ, et al. Expression of the breast cancer metastasis suppressor gene, BRMS1, in
human breast carcinoma: lack of correlation with metastasis to axillary lymph nodes. Tumour
Biol. 2005;26(4):213-216.
doi pubmed - Oshi M, Newman S, Tokumaru Y, Yan L, Matsuyama R, Endo I,
Nagahashi M, et al. Intra-tumoral angiogenesis is associated with inflammation, immune reaction
and metastatic recurrence in breast cancer. Int J Mol Sci. 2020;21(18):6708.
doi pubmed - Pahk K, Joung C, Kim S. Visceral fat metabolic activity evaluated by preoperative 18f-FDG PET/CT significantly affects axillary lymph node metastasis in postmenopausal luminal breast cancer. Sci Rep. 2020;10(1):1348.
- Liu W, Lu X, Shi P, Yang G, Zhou Z, Li W, et al. TNF-α increases breast cancer stem-like cells through up-regulating TAZ expression via the non-canonical NF-κB pathway. Sci Rep. 2020;10(1):1804.
- Katanov C, Lerrer S, Liubomirski Y, Leider-Trejo L, Meshel
T, Bar J, Feniger-Barish R, et al. Regulation of the inflammatory profile of stromal cells in
human breast cancer: prominent roles for TNF-alpha and the NF-kappaB pathway. Stem Cell Res
Ther. 2015;6(1):87.
doi pubmed - Zhang Q, Zhao GS, Yuan XL, Li XH, Yang Z, Cui YF, Guan QL,
et al. Tumor necrosis factor alpha-238G/A polymorphism and risk of breast cancer: an update by
meta-analysis. Medicine (Baltimore). 2017;96(29):e7442.
doi pubmed - Wang Y, Wang X, Zhao H, Liang B, Du Q. Clusterin confers
resistance to TNF-alpha-induced apoptosis in breast cancer cells through NF-kappaB activation
and Bcl-2 overexpression. J Chemother. 2012;24(6):348-357.
doi pubmed - Sethi G, Sung B, Aggarwal BB. TNF: a master switch for
inflammation to cancer. Front Biosci. 2008;13:5094-5107.
doi pubmed - Pileczki V, Braicu C, Gherman CD, Berindan-Neagoe I.
TNF-alpha gene knockout in triple negative breast cancer cell line induces apoptosis.
Int J Mol Sci. 2012;14(1):411-420.
doi pubmed - Mastro AM, Gay CV, Welch DR, Donahue HJ, Jewell J, Mercer R,
DiGirolamo D, et al. Breast cancer cells induce osteoblast apoptosis: a possible contributor to
bone degradation. J Cell Biochem. 2004;91(2):265-276.
doi pubmed - Hauck T, Kadam S, Heinz K, Garcia Peraza M, Schmid R, Kremer
AE, Wolf K, et al. Influence of the autotaxin-lysophosphatidic acid axis on cellular function
and cytokine expression in different breast cancer cell lines. Sci Rep. 2022;12(1):5565.
doi pubmed - Zhang Z, Lin G, Yan Y, Li X, Hu Y, Wang J, Yin B, et al.
Transmembrane TNF-alpha promotes chemoresistance in breast cancer cells. Oncogene.
2018;37(25):3456-3470.
doi pubmed - Al-Qubati AA, Rahmadi M, Widiandani T, Al-Maamari JN, Khotib J. The role of IL-1, IL-6 and TNF-α in breast cancer development and progression. Pharmacy Education. 2024;24(3):32-38.
- Adrian K, Ghaib H, Ali I. Tumour necrosis factor-alpha
levels as predictor factor on clinical response of anthracycline-based neoadjuvant chemotherapy
in locally advance breast cancer patients: experimental research. Ann Med Surg (Lond).
2023;85(4):807-811.
doi pubmed - Efendi R, Ghaib H, Ali I. Association between serum TNF-α level with the incidence of metastases in women with breast cancer in Dr. Soetomo General Hospital, Indonesia. Bali Medical Journal. 2022;11(3):1548-1552.
- Ma Y, Ren Y, Dai ZJ, Wu CJ, Ji YH, Xu J. IL-6, IL-8 and
TNF-alpha levels correlate with disease stage in breast cancer patients. Adv Clin Exp Med.
2017;26(3):421-426.
doi pubmed - Fontvieille E, His M, Biessy C, Navionis AS, Torres-Mejia G,
Angeles-Llerenas A, Alvarado-Cabrero I, et al. Inflammatory biomarkers and risk of breast cancer
among young women in Latin America: a case-control study. BMC Cancer. 2022;22(1):877.
doi pubmed - Berberoglu U, Yildirim E, Celen O. Serum levels of tumor
necrosis factor alpha correlate with response to neoadjuvant chemotherapy in locally advanced
breast cancer. Int J Biol Markers. 2004;19(2):130-134.
doi pubmed - Papadopoulou E, Tripsianis G, Anagnostopoulos K, Tentes I,
Kakolyris S, Galazios G, Sivridis E, et al. Significance of serum tumor necrosis factor-alpha
and its combination with HER-2 codon 655 polymorphism in the diagnosis and prognosis of breast
cancer. Int J Biol Markers. 2010;25(3):126-135.
doi pubmed - Gharib AF, El Askary A, Almehmadi M, Alhuthali HM, Elsawy WH, Allam HH, et al. Association of vitamin D deficiency and inflammatory cytokines with the clinicopathological features of breast cancer in female Saudi patients. Eur J Inflamm. 2022;20.
- Zare Moayedi M, Ahmmadpour F, Rashidi M, Ahmadzadeh A,
Salmasi A, Mohammadzadeh G. The association between mRNA expression of resistin, TNF- alpha,
IL-6, IL-8, and ER-alpha in peripheral blood mononuclear cells and breast cancer.
Turk J Med Sci. 2021;51(3):1345-1353.
doi pubmed - Mohammed AK. Comparison of TNF-alpha and IL-19
concentrations at different stages of breast cancer. J Med Life.
2022;15(6):845-849.
doi pubmed - Darwin N, Qodir N, Umar M, Theodorus. Effects of neo-adjuvant chemotherapy on CD8 + serum levels in local advanced stage breast cancer patients. Sriwijaya Journal of Surgery. 2020;3(1).
- Hashmi AA, Bukhari U, Najam J, Dowlah T, Ali AH, Diwan MA, et al. Luminal B, Human epidermal growth factor receptor 2 (HER2/neu), and triple-negative breast cancers associated with a better chemotherapy response than luminal a breast cancers in postneoadjuvant settings. Cureus. 2023.
- Pratama FM, Ghaib H, Ali I. Correlation between serum Interleukin-6 levels and clinical response to anthracycline-based neoadjuvant chemotherapy regimen in locally advanced breast cancer patients. International Journal of Research and Review. 2021;8(11).
- Maghina J, Chibwae A, Kapesa A, Kiyeti H. Prevalence and associated psychological factors of anxiety and depression among patients with breast cancer receiving chemotherapy at Bugando Medical Centre in Mwanza: the need for interdepartmental management approach [Internet]. 2022. Available from: https://www.researchsquare.com/article/rs-2216985/v1.
- Hayashi T, Fujita K, Matsushita M, Nonomura N. Main
inflammatory cells and potentials of anti-inflammatory agents in prostate cancer. Cancers
(Basel). 2019;11(8).
doi pubmed - Houghton J, Stoicov C, Nomura S, Rogers AB, Carlson J, Li H,
Cai X, et al. Gastric cancer originating from bone marrow-derived cells. Science.
2004;306(5701):1568-1571.
doi pubmed - Liu S, Tan WY, Chen QR, Chen XP, Fu K, Zhao YY, Chen ZW.
Daintain/AIF-1 promotes breast cancer proliferation via activation of the NF-kappaB/cyclin D1
pathway and facilitates tumor growth. Cancer Sci. 2008;99(5):952-957.
doi pubmed - Zaman K, Driscoll R, Hahn D, Werffeli P, Goodman SL, Bauer
J, Leyvraz S, et al. Monitoring multiple angiogenesis-related molecules in the blood of cancer
patients shows a correlation between VEGF-A and MMP-9 levels before treatment and divergent
changes after surgical vs. conservative therapy. Int J Cancer.
2006;118(3):755-764.
doi pubmed - Antoon JW, Lai R, Struckhoff AP, Nitschke AM, Elliott S,
Martin EC, Rhodes LV, et al. Altered death receptor signaling promotes epithelial-to-mesenchymal
transition and acquired chemoresistance. Sci Rep. 2012;2:539.
doi pubmed - Nizamutdinova IT, Lee GW, Lee JS, Cho MK, Son KH, Jeon SJ,
Kang SS, et al. Tanshinone I suppresses growth and invasion of human breast cancer cells,
MDA-MB-231, through regulation of adhesion molecules. Carcinogenesis.
2008;29(10):1885-1892.
doi pubmed - Phi LTH, Sari IN, Yang YG, Lee SH, Jun N, Kim KS, Lee YK, et
al. Cancer Stem Cells (CSCs) in drug resistance and their therapeutic implications in cancer
treatment. Stem Cells Int. 2018;2018:5416923.
doi pubmed - Nassar D, Blanpain C. Cancer stem cells: basic concepts and
therapeutic implications. Annu Rev Pathol. 2016;11:47-76.
doi pubmed
This
article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0
International License, which permits unrestricted non-commercial use, distribution, and
reproduction in any medium, provided the original work is properly cited.
World
Journal of Oncology is published by Elmer Press Inc.