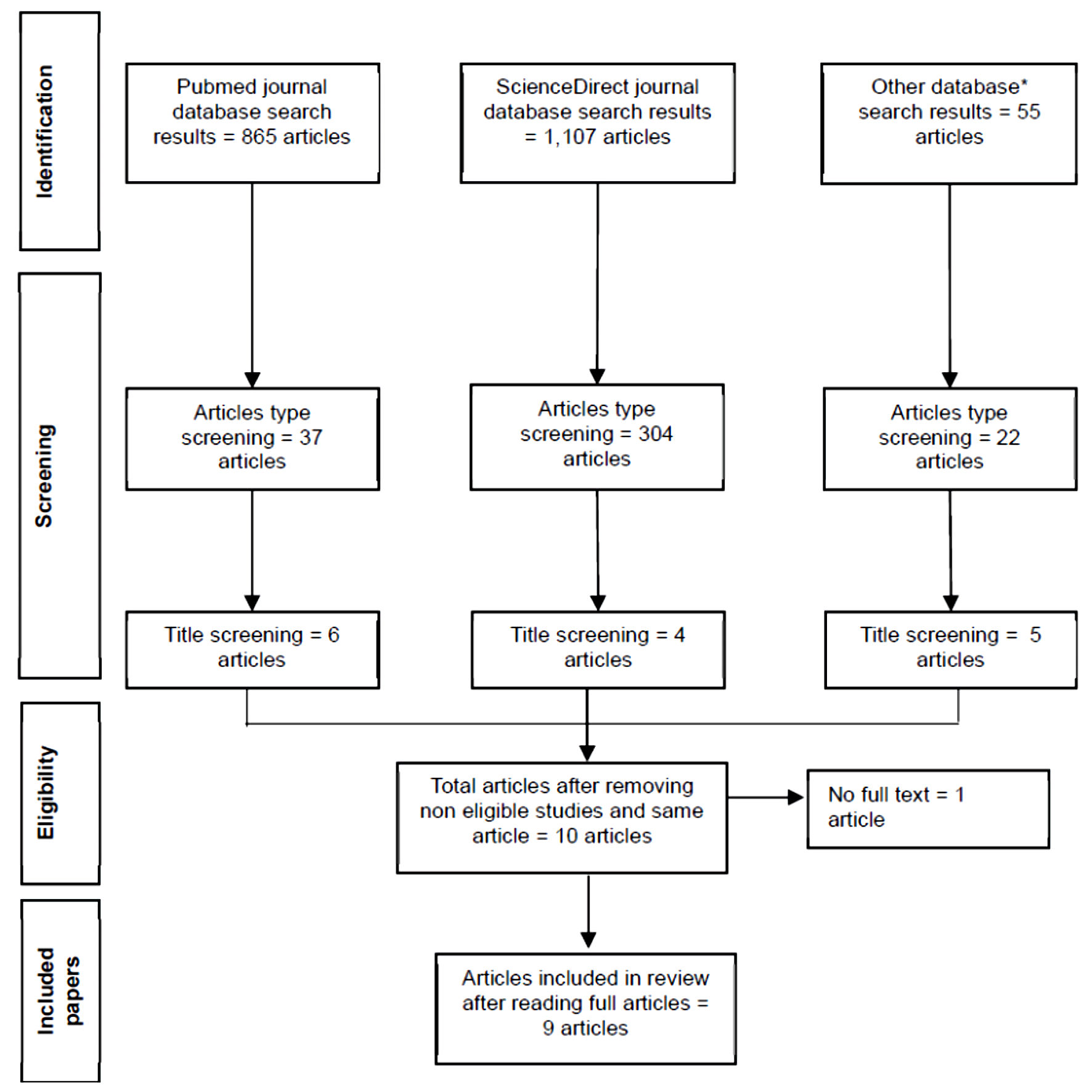
↓ Figure 1. PRISMA flowchart. PRISMA: Preferred
Reporting Items for Systematic Reviews and Meta-Analyses.
| World Journal of Oncology, ISSN 1920-4531 print, 1920-454X online, Open Access |
| Article copyright, the authors; Journal compilation copyright, World J Oncol and Elmer Press Inc |
| Journal website https://wjon.elmerpub.com |
Review
Volume 16, Number 2, April 2025, pages 143-151
Tumor Necrosis Factor-Alpha and Its Association With Breast Cancer: A Systematic Review
Figure

Tables
| Criteria | Inclusion | Exclusion |
|---|---|---|
| TNF-α: tumor necrosis factor-alpha. | ||
| Study design | Observational studies (cross-sectional, case-control, cohort) | Non-observational studies (e.g., reviews, editorials, case reports) |
| Language | Published in English or Indonesian | Published in other languages without an available translation |
| Population | Human studies including breast cancer patients and healthy controls with matched age and relevant demographics | Non-human studies or studies without a well-defined control group/matched population |
| Outcome reporting | Studies that directly assess TNF-α levels about breast cancer with sufficient data | Studies lacking TNF-α measurements or missing relevant data |
| No. | Domain | Study index number | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | ||
| 1 | Comparability of the target group | Low | Low | Low | Unclear | Low | Low | Low | Unclear | High |
| 2 | Target group selection | High | Low | Low | Low | Low | High | Low | Unclear | Low |
| 3 | Confounders | Low | Low | Low | Unclear | Unclear | Low | Low | Low | Unclear |
| 4 | Measurement of exposure | Low | Low | Low | Low | Low | Low | Low | Low | Low |
| 5 | Blinding of assessors | Low | Low | Low | Low | Low | Low | Low | Low | Low |
| 6 | Outcome assessment | Low | Low | Low | Low | Low | Low | Low | Low | Low |
| 7 | Incomplete outcome data | Low | Low | Low | Low | Low | Low | Low | Low | Low |
| 8 | Selective outcome reporting | Low | Low | Low | Low | Low | Low | Low | Low | Low |
| Overall results | High | Low | Low | Some concern | Some concern | High | Low | Some concern | High | |
| No. | Study details | Subject characteristics | Results |
|---|---|---|---|
| TNF-α: tumor necrosis factor-alpha; IQR: interquartile range; AUC: area under the curve; CI: confidence interval; OR: odds ratio; SD: standard deviation. | |||
| 1 | Moaiedi et al, 2021 [30], Iran, cross-sectional | N = 50 (32 women with breast cancer, 18 healthy control) | The mRNA expression of TNF-α was high in breast cancer patients without surgery in comparison to healthy controls, but not significant (P > 0.05), whereas, its mRNA expression of TNF-α was low in patients with surgery in comparison to healthy controls, but not significant (P > 0.05). |
| Median age was 42 years (28 - 60 years) in the breast cancer group and 45 years (36 - 53 years) in the control group. | |||
| 2 | Gharib et al, 2022 [29], Saudi Arabia, case-control | N = 200 (100 women with early or locally advanced breast cancer and 100 healthy women) from 20 to 80 years. | TNF-α was significantly higher in breast cancer patients compared to healthy controls. |
| The mean TNF-α level was 42.15 ± 18.76 pg/mL in the patient group compared to 5.54 ± 2.32 pg/mL in the control group (t = 19.26, P < 0.0001). | |||
| 3 | Papadopoulou et al, 2010 [28], Greece, case-control | N = 101 (56 women with primary breast cancer and 45 healthy women). | The serum levels of TNF-α were significantly higher in patients with primary breast cancer than in the control group (19.18 pg/mL, IQR: 12.04 - 32.51 pg/mL vs. 7.92 pg/mL, IQR: 4.41 - 12.14 pg/mL, P < 0.001). |
| The mean age was 61.3 years (33 - 88 years) in the breast cancer group and 57.2 years (31 - 82 years) in the control group. | The AUC’s diagnostic significance of TNF-α in breast cancer was 0.848 (95% CI: 0.774 - 0.923, P < 0.001). | ||
| Statistically significantly elevated levels of TNF-α were found in larger tumors (P = 0.038), poorly differentiated tumors (P = 0.011), and in patients with more than three positive lymph nodes (P = 0.046). | |||
| A marginal trend towards higher values of TNF-α was found in invasive tumors (P = 0.069) and in advanced-stage carcinomas (P = 0.062). | |||
| The mean survival time was 44 ± 2 months (95% CI: 39 - 48 months) in patients with low levels of TNF-α (≤ 21.55 pg/mL; n = 31) and 35 ± 4 months (95% CI: 28 - 42 months) in patients with high levels of TNF-α (> 21.55 pg/mL; n = 25). | |||
| 4 | Berberoglu et al, 2004 [27], Turkey, case-control | N = 32 (20 women with locally advanced breast cancer and 12 healthy women). | TNF-α in breast cancer patients was higher than healthy group (15.9 ± 0.9 pg/mL vs. 5.8 ± 1.7 pg/mL, P < 0.0001). |
| Median age was 45 years (30 - 75 years) in the breast cancer group and 43 years (37 - 66 years) in the control group. | TNF-α was reduced significantly after chemotherapy both in partial and complete response. | ||
| There was a significant correlation between the type of response and the relative change in TNF-α values (r = -0.62, P = 0.004). | |||
| 5 | Adrian et al, 2023 [23], Indonesia, cross-sectional | N = 38 women with breast cancer who received anthracycline-based neoadjuvant chemotherapy and never received chemotherapy before (20 participants were < 50 years old). | Most participants with high TNF-α levels had a negative response to chemotherapy, while those with low TNF-α levels had a positive response (P < 0.001). |
| The statical analysis showed a significant association between TNF-α levels and the clinical response to chemotherapy, which showed r = -0.606 and P < 0.001. | |||
| 6 | Efendi et al, 2022 [24], Indonesia, cross-sectional | N = 50 women with breast cancer | There is a statistically significant association between serum TNF-α levels and the incidence of breast cancer metastases (P = 0.009) |
| The mean age was 48.8 years (29 - 65 years). | |||
| 7 | Fontvieille et al, 2022 [26], France, case-control | N = 906 (453 women with breast cancer and 453 control from the general population). | In the fully adjusted model, TNF-α was positively associated with breast cancer risk (OR per SD increment = 1.32 (1.11 - 1.58); OR Q4 vs. Q1= 2.03 (1.26 - 3.26), P= 0.006). |
| The case and control groups have a mean age of 38.7 years | |||
| 8 | Ma et al, 2017 [25], China, case-control | N = 140 (110 women with breast cancer and 30 healthy women). | The serum TNF-α levels of stage III carcinoma patients were significantly higher than those in the healthy controls (P < 0.001). |
| Age range from 35 to 65 years in the breast cancer group and 28 to 57 years in the healthy group. | Serum TNF-α levels also correlated with clinical tumor stage and lymph node metastasis (P < 0.001). | ||
| 9 | Mohammed et al, 2022 [31], Iraq, case-control | N = 90 (59 women with breast cancer and 31 healthy women). | Individuals with ductal carcinoma showed significant differences (P < 0.001) in the mean levels of the TNF-α compared with healthy groups |
| The mean age was 37.4 years in the breast cancer group and 34.7 years in the control group (25 - 61 years). | There is a significantly increased level of TNF-α in lymph node metastasis compared to healthy groups. | ||
| There is no significant difference in the levels of TNF-α between the non-metastasis and metastasis groups. | |||