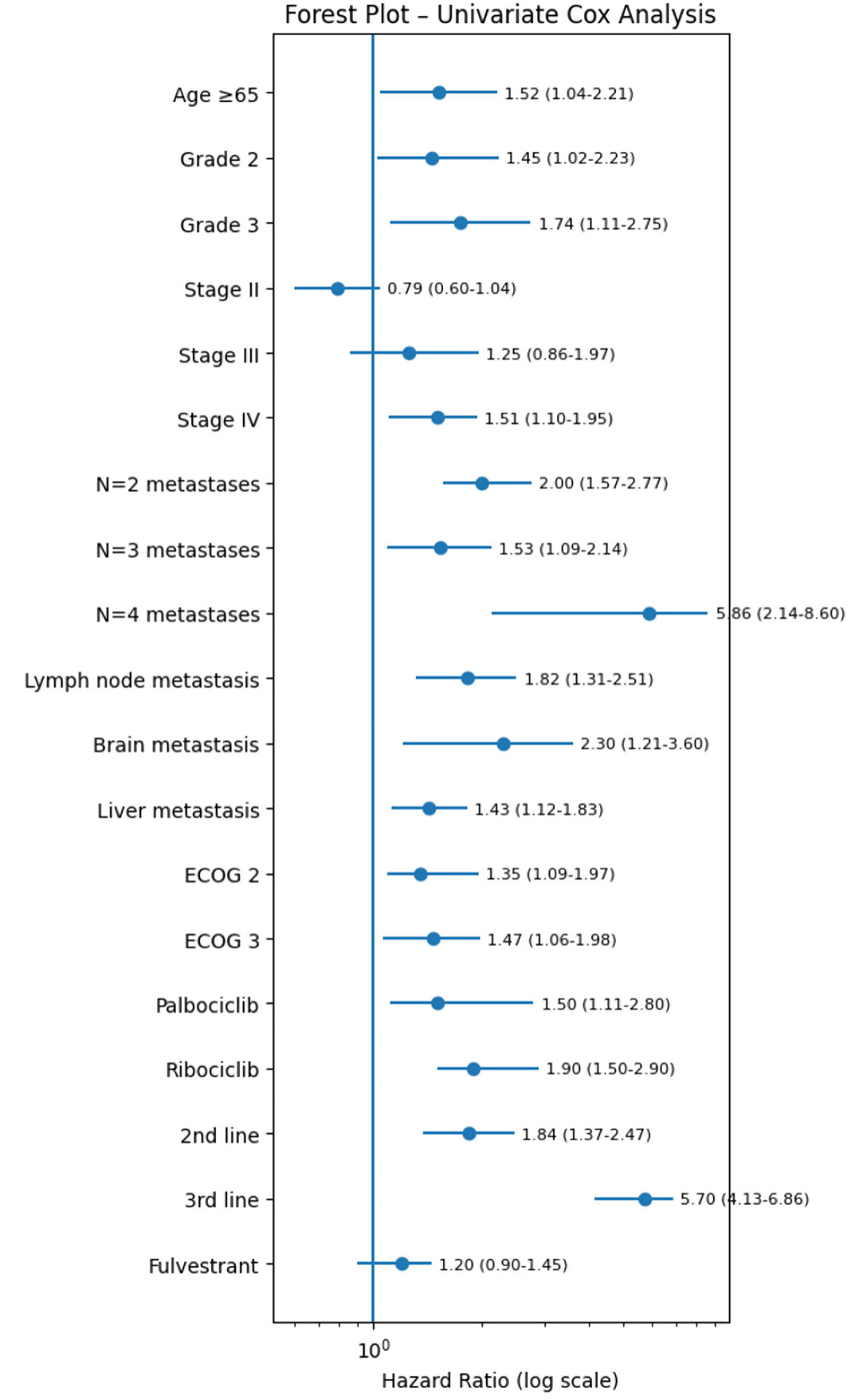
↓ Figure 1. Univariate forest plot model for mPFS of patients receiving CDK4/6i. CDK4/6i: cyclin-dependent kinase 4 and 6 inhibitors; ECOG: Eastern Cooperative Oncology Group; PFS: progression-free survival.
| World Journal of Oncology, ISSN 1920-4531 print, 1920-454X online, Open Access |
| Article copyright, the authors; Journal compilation copyright, World J Oncol and Elmer Press Inc |
| Journal website https://wjon.elmerpub.com |
Original Article
Volume 17, Number 3, June 2026, pages 380-393
Real-World Data on the Use of Cyclin-Dependent Kinase 4/6 Inhibitors in Hormone Receptor–Positive/Human Epidermal Growth Factor Receptor 2–Negative Advanced/Metastatic Breast Cancer
Figures

Tables
| Characteristica | Overall (n = 269) | Palbociclib (n = 149) | Ribociclib (n = 103) | Abemaciclib (n = 17) | P valueb |
|---|---|---|---|---|---|
| aN (%), unless otherwise specified. bKruskal-Wallis rank sum test; Fisher’s exact test. cMean ± SD (range). dPatients can have multiple metastatic sites. ECOG: Eastern Cooperative Oncology Group; SD: standard deviation; a/mBC: advanced/metastatic breast cancer; CDK4/6i: cyclin-dependent kinase 4 and 6 inhibitors; HER2–: human epidermal growth factor receptor 2–negative; HR+: hormone receptor–positive. | |||||
| Age (years) at initial diagnosisc | 51 ± 11 (29–82) | 49 ± 9 (30–75) | 55 ± 13 (29–82) | 51 ± 12 (35–70) | 0.002 |
| Breast cancer orientation at initial diagnosis | 0.4 | ||||
| Left breast | 177 (66%) | 102 (68%) | 66 (64%) | 9 (53%) | |
| Right breast | 92 (34%) | 47 (32%) | 37 (36%) | 8 (47%) | |
| Breast cancer subtype at initial diagnosis | 0.2 | ||||
| Ductal carcinoma | 190 (71%) | 111 (74%) | 66 (64%) | 13 (76%) | |
| Lobular carcinoma | 79 (29%) | 38 (26%) | 37 (36%) | 4 (24%) | |
| Breast cancer grade at initial diagnosis | 0.6 | ||||
| 1 | 24 (8.9%) | 15 (10%) | 8 (7.8%) | 1 (5.9%) | |
| 2 | 145 (54%) | 80 (54%) | 53 (51%) | 12 (71%) | |
| 3 | 100 (37%) | 54 (36%) | 42 (41%) | 4 (24%) | |
| Breast cancer stage at initial diagnosis | 0.001 | ||||
| Early stage I | 23 (8.6%) | 3 (2.0%) | 17 (17%) | 3 (18%) | |
| Early stage II | 112 (42.1%) | 79 (53%) | 30 (28.9%) | 3 (18%) | |
| Locally advanced stage III | 36 (13%) | 22 (15%) | 9 (8.7%) | 5 (29%) | |
| De novo metastatic stage IV | 98 (36%) | 45 (30%) | 47 (46%) | 6 (35%) | |
| Menopausal status at time of CDK4/6i treatment | 0.5 | ||||
| Postmenopausal | 237 (88%) | 134 (90%) | 88 (85%) | 15 (88%) | |
| Premenopausal | 32 (12%) | 15 (10%) | 15 (15%) | 2 (12%) | |
| ECOG performance status at time of CDK4/6i treatment | 0.002 | ||||
| 0 | 27 (10%) | 14 (9%) | 11 (10%) | 2 (10%) | |
| 1 | 89 (33%) | 47 (32%) | 38 (37%) | 4 (25%) | |
| 2 | 78 (29%) | 38 (26%) | 33 (32%) | 7 (41%) | |
| 3 | 75 (28%) | 49 (33%) | 22 (21%) | 4 (24%) | |
| Metastatic sites at time of CDK4/6i treatmentd | |||||
| Bones | 75 (28%) | 94 (63%) | 68 (66%) | 16 (94%) | 0.038 |
| Lungs | 115 (43%) | 36 (24%) | 30 (29%) | 9 (53%) | 0.043 |
| Lymph nodes | 178 (66%) | 15 (10%) | 26 (25%) | 4 (24%) | 0.003 |
| Brain | 45 (17%) | 31 (21%) | 23 (22%) | 1 (5.9%) | 0.3 |
| Liver | 55 (20%) | 55 (37%) | 54 (52%) | 6 (35%) | 0.041 |
| Overall (n = 269) | Palbociclib (n = 149) | Ribociclib (n = 103) | Abemaciclib (n = 17) | P valuea | |
|---|---|---|---|---|---|
| aKruskal-Wallis rank sum test; Fisher’s exact test. bDaily dose level 1: 125 mg for palbociclib; 600 mg for ribociclib; 300 mg for abemaciclib (FDA-recommended starting daily dose). Daily dose level 2: 100 mg for palbociclib; 400 mg for ribociclib; 150 mg for abemaciclib. Daily dose level 3: 75 mg for palbociclib; 200 mg for ribociclib. CDK4/6i: cyclin-dependent kinase 4/6 inhibitor; SD: standard deviation; FDA: FDA: Food and Drug Administration. | |||||
| Line of therapy, n (%) | < 0.001 | ||||
| 1 | 182 (68%) | 113 (76%) | 61 (59%) | 8 (47%) | |
| 2 | 66 (25%) | 31 (21%) | 32 (31%) | 3 (18%) | |
| 3 | 21 (7.8%) | 5 (3.4%) | 10 (9.7%) | 6 (35%) | |
| Starting total daily dose of CDK4/6i, n (%)b | < 0.001 | ||||
| 75 mg | 1 (0.4%) | 1 (0.7%) | 0 | 0 | |
| 100 mg | 28 (10%) | 28 (19%) | 0 | 0 | |
| 125 mg | 120 (45%) | 120 (81%) | 0 | 0 | |
| 150 mg | 3 (1.1%) | 0 | 0 | 3 (18%) | |
| 200 mg | 2 (0.7%) | 0 | 2 (1.9%) | 0 | |
| 300 mg | 14 (5.2%) | 0 | 0 | 14 (82%) | |
| 400 mg | 20 (7.4%) | 0 | 20 (19%) | 0 | |
| 600 mg | 81 (30%) | 0 | 81 (79%) | 0 | < 0.001 |
| Ending total daily dose of CDK4/6i, n (%)b | |||||
| 75 mg | 10 (3.7%) | 10 (6.7%) | 0 | 0 | |
| 100 mg | 73 (27%) | 73 (49%) | 0 | 0 | |
| 125 mg | 66 (25%) | 66 (44%) | 0 | 0 | |
| 150 mg | 7 (2.6%) | 0 | 0 | 7 (41%) | |
| 200 mg | 11 (4.1%) | 0 | 11 (11%) | 0 | |
| 300 mg | 10 (3.7%) | 0 | 0 | 10 (59%) | |
| 400 mg | 34 (13%) | 0 | 34 (33%) | 0 | |
| 600 mg | 58 (22%) | 0 | 58 (56%) | 0 | |
| Endocrine therapy given in combination with CDK4/6i, n (%) | < 0.001 | ||||
| Fulvestrant | 79 (29%) | 33 (22%) | 36 (35%) | 10 (59%) | |
| Letrozole | 190 (71%) | 116 (78%) | 67 (65%) | 7 (41%) | |
| Line of endocrine therapy in combination with CDK4/6i, n (%) | < 0.001 | ||||
| 1 | 92 (34%) | 44 (30%) | 43 (45%) | 5 (31%) | |
| 2 | 136 (51%) | 98 (66%) | 33 (35%) | 5 (31%) | |
| 3 | 31 (12%) | 6 (4.1%) | 19 (20%) | 6 (38%) | |
| CDK4/6i dose reduction, n (%) | 99 (37%) | 63 (42%) | 32 (31%) | 4 (24%) | |
| Time to CDK4/6i dose reduction (in days) | < 0.001 | ||||
| Number of observations | 269 | 149 | 103 | 17 | |
| Mean (SD) | 79 (39) | 84 (29) | 67 (17) | 38 (15) | |
| CDK4/6i dose discontinuation, n (%) | 180 (67%) | 105 (70%) | 69 (67%) | 6 (35%) | |
| Time to CDK4/6i discontinuation (in days) | < 0.001 | ||||
| Number of observations | 269 | 149 | 103 | 17 | |
| Mean (SD) | 845 (153) | 942 (93) | 755 (95) | 541 (134) | |
| Overall (n = 269) | Palbociclib (n = 149) | Ribociclib (n = 103) | Abemaciclib (n = 17) | P valuea | |
|---|---|---|---|---|---|
| Follow-up time stopped at the time of CDK discontinuation or end of the study period, whichever came first. aFisher’s exact test. AE: adverse event; a/mBC: advanced/metastatic breast cancer; CDK4/6i: cyclin-dependent kinase 4 and 6 inhibitors; HER2–: human epidermal growth factor receptor 2–negative; HR+: hormone receptor–positive; AST: aspartate aminotransferase; ALT: alanine aminotransferase. | |||||
| Experienced ≥ 1 protocol-defined AEs, n (%) | |||||
| ALT/AST increase grade 1 | 15 (5.6%) | 0 | 13 (13%) | 2 (12%) | 6.81 × 10–6 |
| ALT/AST increase grade 3 | 21 (7.8%) | 0 | 21 (20%) | 0 | 2.03 × 10–9 |
| Diarrhea grade 1 | 52 (19%) | 38 (26%) | 14 (14%) | 0 | 0.005 |
| Diarrhea grade 2 | 19 (7.1%) | 11 (7.4%) | 0 | 8 (47%) | 2.52 × 10–8 |
| Diarrhea grade 3 | 4 (1.5%) | 0 | 0 | 4 (24%) | 1.12 × 10–5 |
| Neutropenia grade 1 | 35 (13%) | 13 (8.7%) | 19 (18%) | 3 (18%) | 0.048 |
| Neutropenia grade 2 | 8 (3.0%) | 0 | 8 (7.8%) | 0 | 0.001 |
| Neutropenia grade 3 | 47 (17%) | 44 (30%) | 3 (2.9%) | 0 | 6.01 × 10–9 |
| Neutropenia grade 4 | 19 (7.1%) | 19 (13%) | 0 | 0 | 7.35 × 10–5 |
| Prolonged QT interval | 5 (1.9%) | 0 | 5 (4.9%) | 0 | 0.025 |
| Thrombocytopenia | 13 (4.8%) | 13 (8.7%) | 0 | 0 | 0.002 |
| Pneumonia grade 1 | 8 (3.0%) | 0 | 8 (7.8%) | 0 | 0.001 |
| Pneumonia grade 2 | 14 (5.2%) | 11 (7.4%) | 3 (2.9%) | 0 | 0.24 |
| Erythematous rash | 9 (3.3%) | 0 | 9 (8.7%) | 0 | 4.32 × 10–4 |
| Shortness of breath | 3 (1.1%) | 3 (2.0%) | 0 | 0 | 0.4 |
| Dose reduction due to AE, n (%) | 99 (37%) | 63 (42%) | 32 (28%) | 4 (24%) | 1.89 × 10–4 |
| ALT/AST increase grade 3 | 21 (7.8%) | 0 | 21 (20%) | 0 | 2.03 × 10–9 |
| Neutropenia grade 3 | 47 (17%) | 44 (30%) | 3 (2.9%) | 0 | 6.01 × 10–9 |
| Neutropenia grade 4 | 19 (7.1%) | 19 (13%) | 0 | 0 | 7.35 × 10–5 |
| Prolonged QT interval | 5 (1.9%) | 0 | 5 (4.9%) | 0 | 0.025 |
| Pneumonia grade 2 | 3 (1.2%) | 1 (0.6%) | 2 (1.9%) | 0 | 0.24 |
| Diarrhea grade 3 | 4 (1.6%) | 0 | 0 | 4 (24%) | 1.6 × 10–5 |
| CDK4/6i discontinuation due to AE, n (%) | |||||
| Neutropenia grade 4 | 3 (1.1%) | 3 (2.0%) | 0 | 0 | 0.4 |
| Univariate analysis | Multivariate analysis | |||||||
|---|---|---|---|---|---|---|---|---|
| Reference | No reference | Median PFS (months) | HR | 95% CI | P value | HR | 95% CI | P value |
| HR: hazard ratio; BC: breast cancer; CDK4/6i: cyclin-dependent kinase 4 and 6 inhibitors; CI: confidence interval; ECOG: Eastern Cooperative Oncology Group; PFS: progression-free survival; ref: reference. | ||||||||
| Age at diagnosis (ref: < 65 years) | ≥ 65 years | 29.37 vs 25.4 | 1.52 | 1.04–2.21 | 0.03 | 1.49 | 1.15–2.54 | 0.022 |
| Menopausal status (ref: postmenopause) | Premenopause | 28.43 vs 26.37 | 1.01 | 0.65–1.57 | 0.96 | - | - | - |
| Breast cancer histology at initial diagnosis (ref: ductal carcinoma) | Lobular carcinoma | 29.37 vs 27.4 | 0.94 | 0.72–1.23 | 0.67 | - | - | - |
| Breast cancer grade at initial diagnosis (ref: grade 1) | Grade 2 | 31.43 vs 28.33 | 1.45 | 1.02–2.23 | 0.04 | 1.06 | 0.7–1.9 | 0.8 |
| Grade 3 | 31.43 vs 26.45 | 1.74 | 1.11–2.75 | 0.016 | 1.52 | 1.15–2.28 | 0.032 | |
| Breast cancer stage at initial diagnosis (ref: stage I) | Stage II | 31.4 vs 29.5 | 0.79 | 0.6–1.04 | 0.1 | - | - | - |
| Stage III | 31.4 vs 27.2 | 1.25 | 0.86–1.97 | 0.42 | - | - | - | |
| De novo metastatic stage IV | 31.4 vs 26.4 | 1.51 | 1.1–1.95 | 7.38 × 10–5 | 1.87 | 1.18–2.9 | 0.04 | |
| Number of metastatic sites at time of CDK4/6i treatment (ref: n = 1) | N = 2 | 31.42 vs 27.37 | 2 | 1.57–2.77 | 4.51 × 10–3 | 1.4 | 0.6–4.07 | 0.52 |
| N = 3 | 31.42 vs 26.33 | 1.53 | 1.09–2.14 | 0.014 | 1.9 | 1.23–4.03 | 0.059 | |
| N = 4 | 31.42 vs 23.30 | 5.86 | 2.14–8.6 | 1.41 × 10–6 | 2.35 | 1.37–5.61 | 0.045 | |
| Bone metastasis (ref: no) | Yes | 30.4 vs 26.37 | 1.16 | 0.9–1.49 | 0.26 | - | - | - |
| Lung metastasis (ref: no) | Yes | 29.38 vs 27.33 | 1.17 | 0.89–1.55 | 0.25 | - | - | - |
| Lymph node metastasis (ref: no) | Yes | 30.37 vs 25.33 | 1.82 | 1.31–2.51 | 3.17 × 10–4 | 1.76 | 1.14–4.03 | 0.033 |
| Brain metastasis (ref: no) | Yes | 31.8 vs 23.10 | 2.3 | 1.21–3.6 | 0.02 | 2.1 | 0.9–2.19 | 0.175 |
| Liver metastasis (ref: no) | Yes | 30.47 vs 26.37 | 1.43 | 1.12–1.83 | 4.09 × 10–3 | 1.32 | 0.8–2.94 | 0.2 |
| ECOG performance at time of CDK4/6i treatment (ref: ECOG 1) | ECOG 2 | 31.4 vs 29.9 | 1.35 | 1.09–1.97 | 0.04 | 1.18 | 0.71–2.46 | 0.73 |
| ECOG 3 | 31.4 vs 27.4 | 1.47 | 1.06–1.98 | 0.011 | 1.25 | 1.06–1.97 | 3.09 × 10–3 | |
| CDK4/6i (ref: abemaciclib) | Palbociclib | 20.27 vs 31.43 | 1.5 | 1.11–2.8 | 4.96 × 10–8 | 0.8 | 0.43–0.9 | 2.7 × 10–4 |
| Ribociclib | 20.27 vs 25.37 | 1.9 | 1.5–2.9 | 7.59 × 10–5 | 0.92 | 0.62–1.67 | 0.48 | |
| Line of CDK4/6i treatment (ref: first line) | Second line | 31.5 vs 26.3 | 1.84 | 1.37–2.47 | 5.29 × 10–5 | 1.17 | 1.08–3.46 | 2.39 × 10–3 |
| Third line | 31.5 vs 21.27 | 5.7 | 4.13–6.86 | 1.34 × 10–4 | 5.38 | 2.04–7.08 | 8.06 × 10–3 | |
| Partner drug–endocrine therapy (ref: letrozole) | Fulvestrant | 31.4 vs 28.37 | 1.2 | 0.9–1.45 | 0.29 | - | - | - |
| Univariate analysis | Multivariate Analysis | |||||||
|---|---|---|---|---|---|---|---|---|
| Reference | No reference | Median OS (months) | HR | 95% CI | P value | HR | 95% CI | P value |
| HR: hazard ratio; BC: breast cancer; CDK4/6i: cyclin-dependent kinase 4 and 6 inhibitors; CI: confidence interval; ECOG: Eastern Cooperative Oncology Group; mOS: median overall survival; ref: reference. | ||||||||
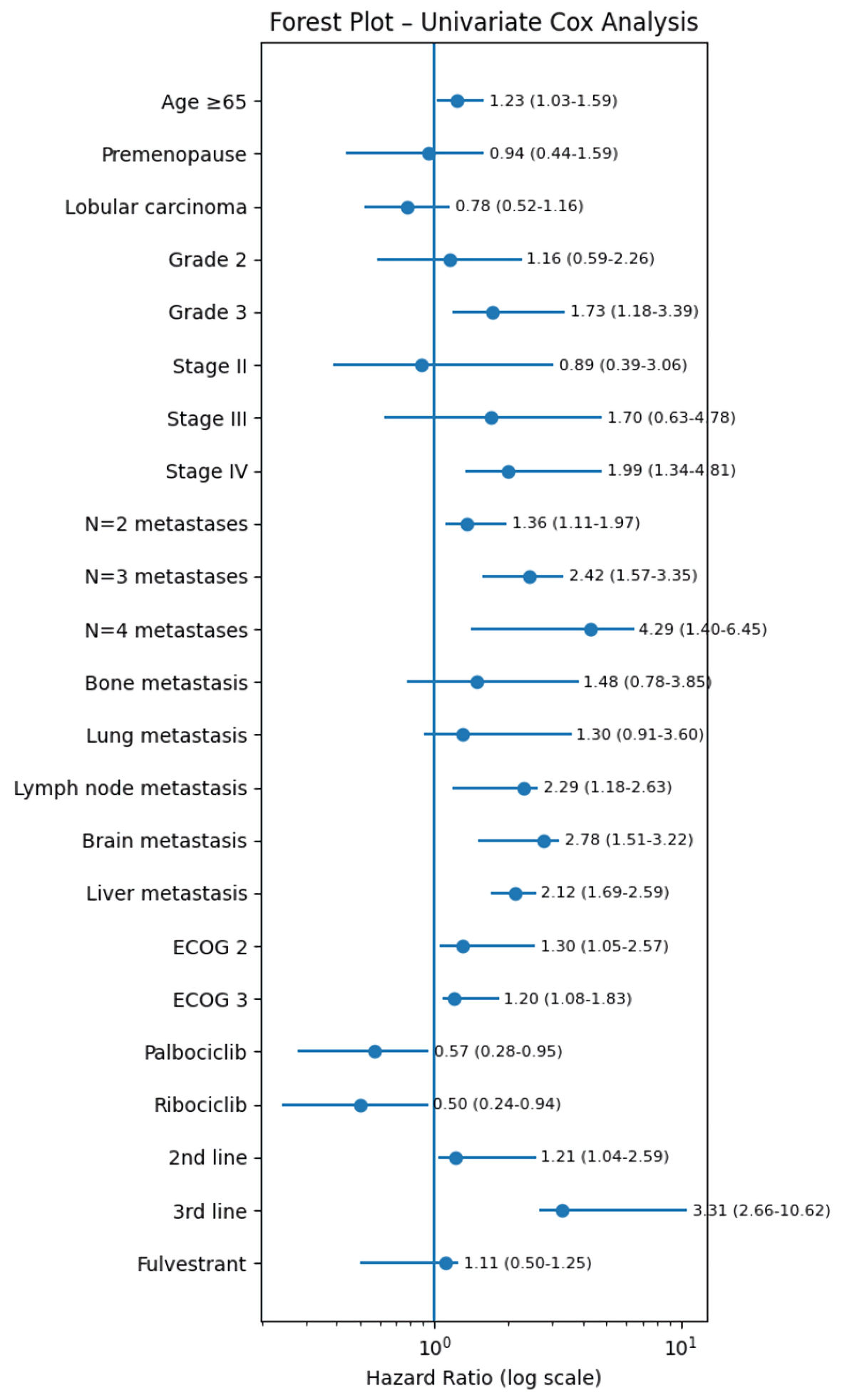
| Age at diagnosis (ref: < 65 years) | ≥ 65 years | 55.23 vs 51.7 | 1.23 | 1.03–1.59 | 0.042 | 1.58 | 1.26–2.69 | 0.034 |
| Menopausal status (ref: postmenopause) | Premenopause | 56.8 vs 53.67 | 0.94 | 0.44–1.59 | 0.68 | - | - | - |
| Breast cancer histology at initial diagnosis (ref: ductal carcinoma) | Lobular carcinoma | 55.8 vs 53.7 | 0.78 | 0.52–1.16 | 0.22 | - | - | - |
| Breast cancer grade at initial diagnosis (ref: grade 1) | Grade 2 | 60.32 vs 55.8 | 1.16 | 0.59–2.26 | 0.66 | - | - | - |
| Grade 3 | 60.32 vs 51.2 | 1.73 | 1.18–3.39 | 0.02 | 1.17 | 1.03–2.79 | 0.037 | |
| Breast cancer stage at initial diagnosis (ref: stage I) | Stage II | 58.83 vs 44.43 | 0.89 | 0.39–3.06 | 0.23 | |||
| Stage III | 58.83 vs 40.08 | 1.7 | 0.63–4.78 | 0.36 | ||||
| De novo metastatic stage IV | 58.83 vs 33.4 | 1.99 | 1.34–4.81 | 2.6 × 10–4 | 1.96 | 1.02–3.4 | 0.03 | |
| Number of metastatic sites at time of CDK4/6i treatment (ref: n = 1) | N = 2 | 56.73 vs 53.77 | 1.36 | 1.11–1.97 | 0.03 | 1.28 | 0.6–1.8 | 0.73 |
| N = 3 | 56.73 vs 50.6 | 2.42 | 1.57–3.35 | 0.6 × 10–3 | 2.15 | 1.13–4.03 | 0.02 | |
| N = 4 | 56.73 vs 46.67 | 4.29 | 1.4–6.45 | 4.29 × 10–5 | 2.56 | 1.73–6.81 | 0.039 | |
| Bone metastasis (ref: no) | Yes | 55.46 vs 50.28 | 1.48 | 0.78–3.85 | 0.12 | - | - | - |
| Lung metastasis (ref: no) | Yes | 58.8 vs 51.72 | 1.3 | 0.91–3.6 | 0.29 | - | - | - |
| Lymph node metastasis (ref: no) | Yes | 60.77 vs 50.73 | 2.29 | 1.18–2.63 | 0.003 | 1.53 | 1.28–3.06 | 0.017 |
| Brain metastasis (ref: no) | Yes | 57.4 vs 48.67 | 2.78 | 1.51–3.22 | 0.001 | 2.1 | 1.34–2.19 | 0.04 |
| Liver metastasis (ref: no) | Yes | 56.8 vs 53.73 | 2.12 | 1.69–2.59 | 1.6 × 10–3 | 1.32 | 0.8–2.94 | 0.36 |
| ECOG performance at time of CDK4/6i treatment (ref: ECOG 1) | ECOG 2 | 62.87 vs 54.77 | 1.3 | 1.05 – 2.57 | 0.008 | 2.07 | 0.63–4.82 | 0.39 |
| ECOG 3 | 62.87 vs 50.68 | 1.2 | 1.08–1.83 | 0.042 | 1.39 | 1.06–1.99 | 3.1 × 10–3 | |
| CDK4/6i (ref: abemaciclib) | Palbociclib | 43.63 vs 59.77 | 0.57 | 0.28–0.95 | 0.01 | 0.37 | 0.2–0.8 | 2.35 × 10–5 |
| Ribociclib | 43.63 vs 53.73 | 0.5 | 0.24–0.94 | 0.04 | 0.75 | 0.46–2.93 | 0.61 | |
| Line of CDK4/6i treatment (ref: first line) | Second line | 57.8 vs 47.43 | 1.21 | 1.04–2.59 | 0.0033 | 2.79 | 1.98–7.36 | 0.81 × 10–3 |
| Third line | 57.8 vs 42.67 | 3.31 | 2.66–10.62 | 2.62 × 10–4 | 4.21 | 2.01–9.08 | 1.04 × 10–4 | |
| Partner drug–endocrine therapy (ref: letrozole) | Fulvestrant | 58.33 vs 52.6 | 1.11 | 0.5–1.25 | 0.37 | - | - | - |