Figures
↓ Figure 1. Overview of the bidirectional
two-sample Mendelian randomization (MR) study design. Publicly available genome-wide association study
(GWAS) summary statistics from European-ancestry populations were used for eight prevalent autoimmune
diseases (AIDs)—rheumatoid arthritis, systemic lupus erythematosus, ankylosing spondylitis, gout,
multiple sclerosis, celiac disease, eczema, and asthma—and for colorectal cancer (CRC), with CRC
outcome data obtained from a GWAS meta-analysis (Fernandez-Rozadilla et al, 2023 [30]). For each AID (as
the exposure) and in the reverse analysis with CRC as the exposure, we selected genetic instrumental
variables (IVs) using standard MR criteria (genome-wide significant association with the exposure,
linkage disequilibrium clumping to ensure independence, and harmonization of effect alleles across
exposure and outcome datasets). Harmonized SNP–exposure and SNP–outcome associations were
then analyzed in both directions to estimate causal effects. The inverse-variance weighted (IVW) method
was used as the primary estimator, complemented by weighted median and MR-Egger regression to assess
robustness under different assumptions. Multiple sensitivity analyses were performed to evaluate
heterogeneity and potential horizontal pleiotropy, including Cochran’s Q statistic, MR-Egger
intercept test, MR-pleiotropy residual sum and outlier (MR-PRESSO), leave-one-out analysis, funnel plot
inspection, and Causal Analysis Using Summary Effect Estimates (CAUSE). All procedures and reporting
followed the STROBE-MR guideline. SNP: single nucleotide polymorphism.
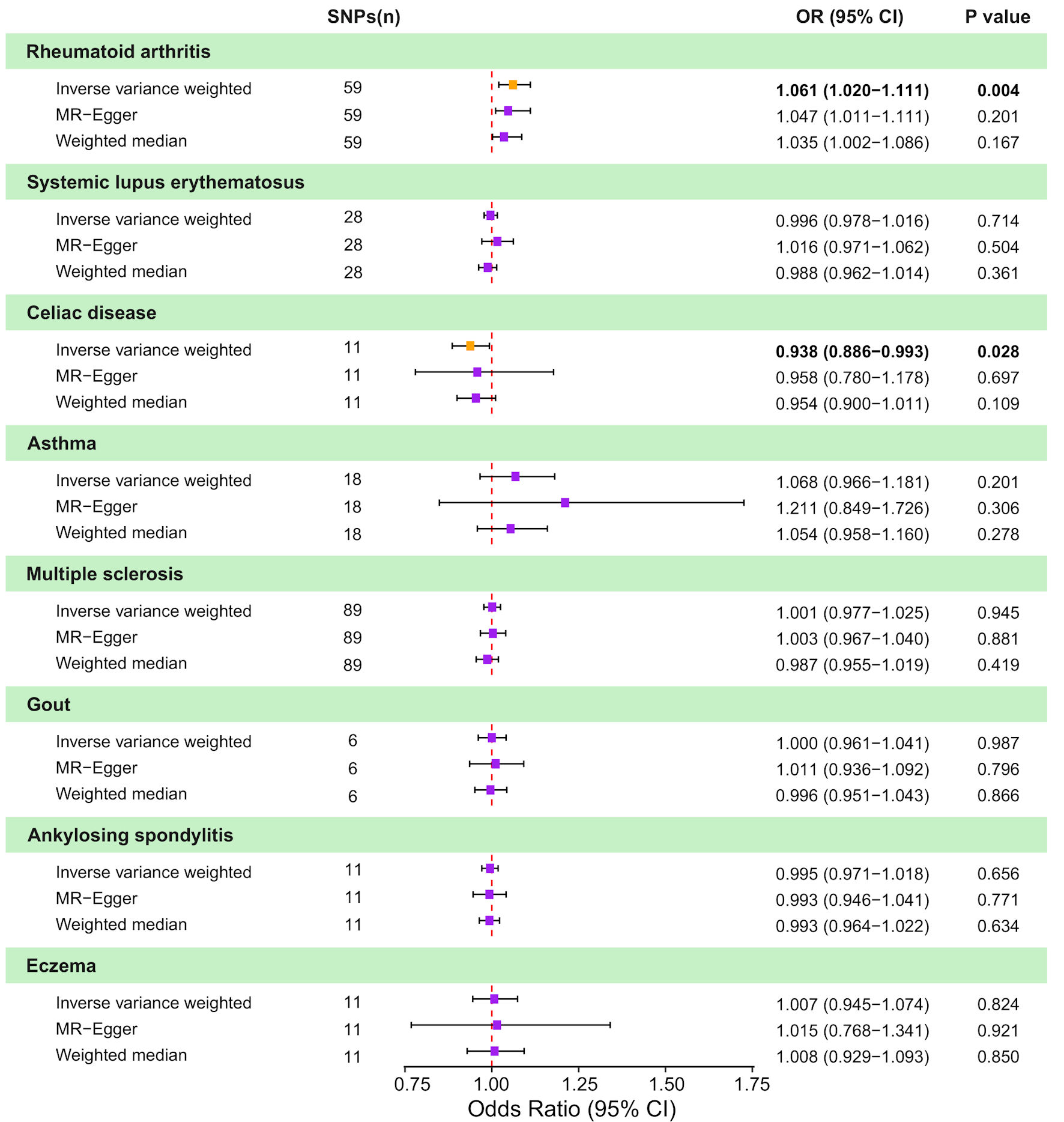
↓ Figure 2. Causal effects of genetically
predicted autoimmune diseases (AIDs) on colorectal cancer (CRC) risk in two-sample Mendelian
randomization (MR) analyses. Forest plot shows odds ratio (OR) and 95% confidence interval (CI) for CRC
per genetically predicted liability to eight AIDs: rheumatoid arthritis, systemic lupus erythematosus,
celiac disease, asthma, multiple sclerosis, gout, ankylosing spondylitis, and eczema. For each AID,
estimates are presented for the inverse-variance weighted (IVW) method, MR-Egger regression, and the
weighted median approach, along with the corresponding number of instrumental SNPs and P values. Squares
represent estimates and horizontal lines indicate 95% CIs; the vertical dashed line denotes the null
effect (OR = 1.0). SNP: single nucleotide polymorphism.
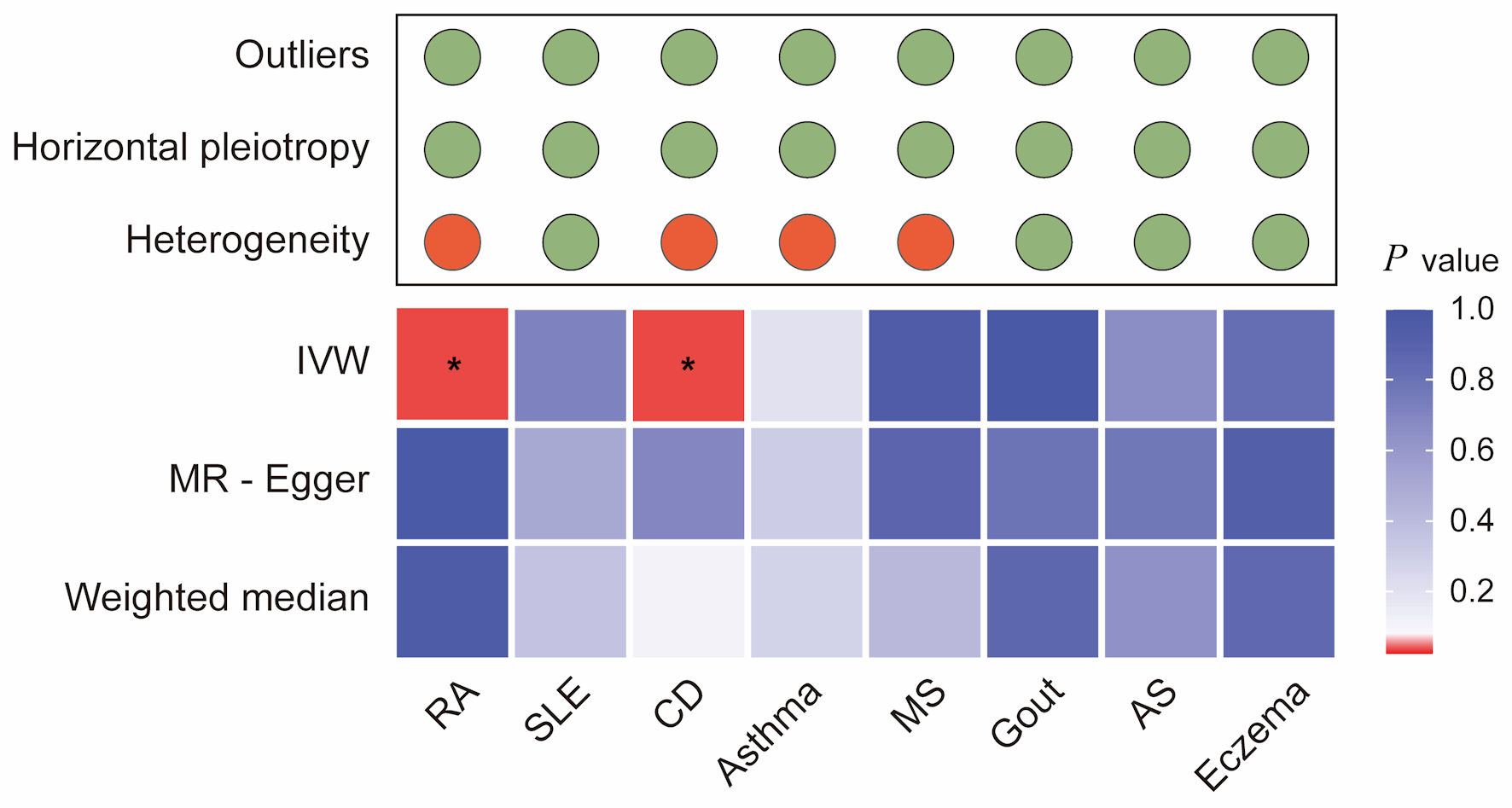
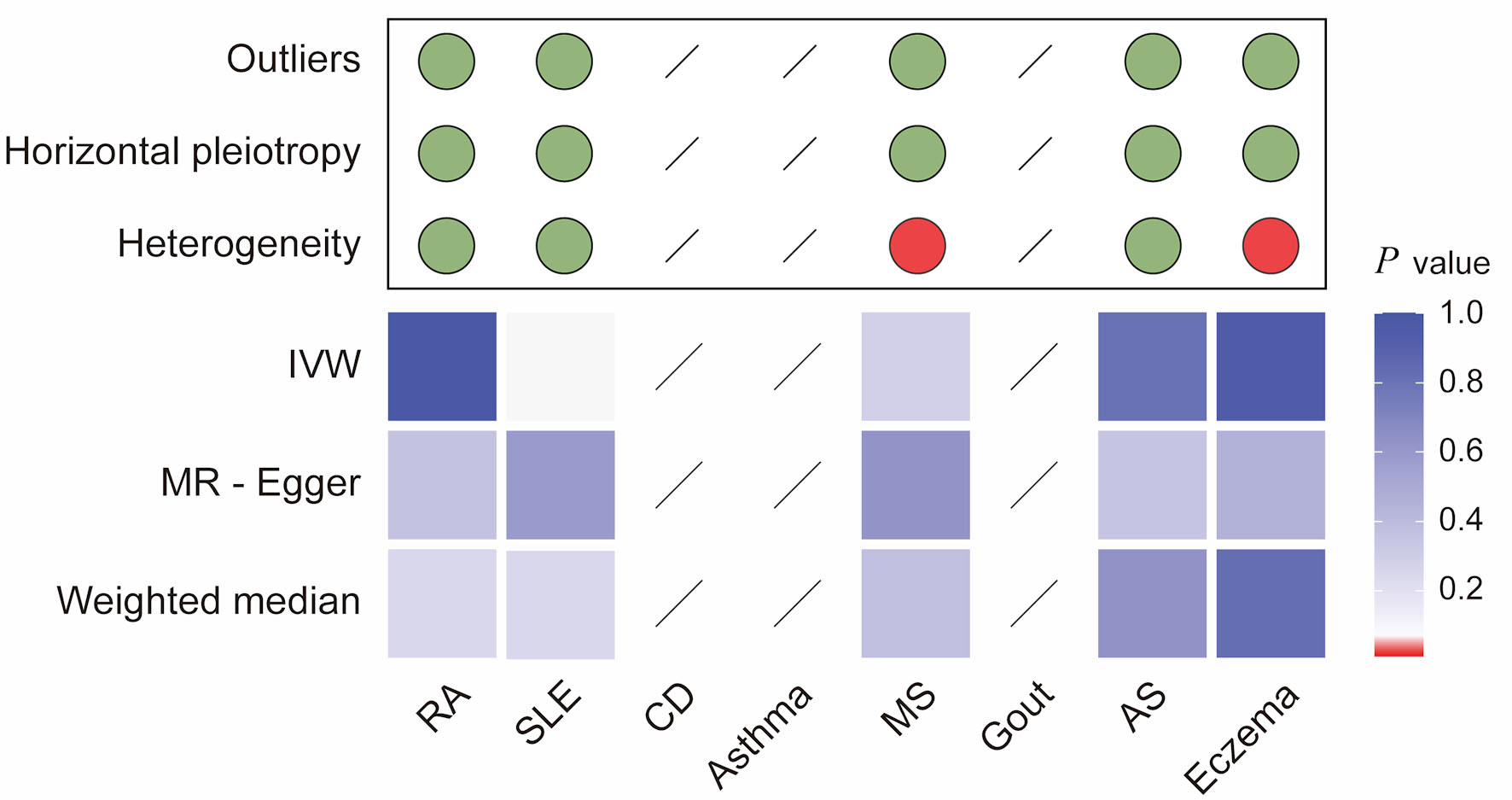
↓ Figure 3. Integrated summary of Mendelian
randomization (MR) estimates and sensitivity analyses for autoimmune diseases and colorectal cancer. The
heatmap shows the strength of evidence from three MR methods (IVW, MR-Egger, and weighted median) for
testing whether genetic susceptibility to eight autoimmune diseases (RA, SLE, CD, asthma, MS, gout, AS,
and eczema) is causally related to colorectal cancer (CRC). The red boxes indicate the existence of a
statistically significant causal relationship, whereas the blue boxes represent a lack of evidence for a
causal relationship. The upper panel summarizes the results of sensitivity analyses, with green circles
indicating no evidence and red circles indicating the presence of outliers, horizontal pleiotropy, or
heterogeneity. *P value < 0.05. RA: rheumatoid arthritis; SLE: systemic lupus erythematosus; AS:
ankylosing spondylitis; CD: celiac disease; IVW: inverse-variance weighted.
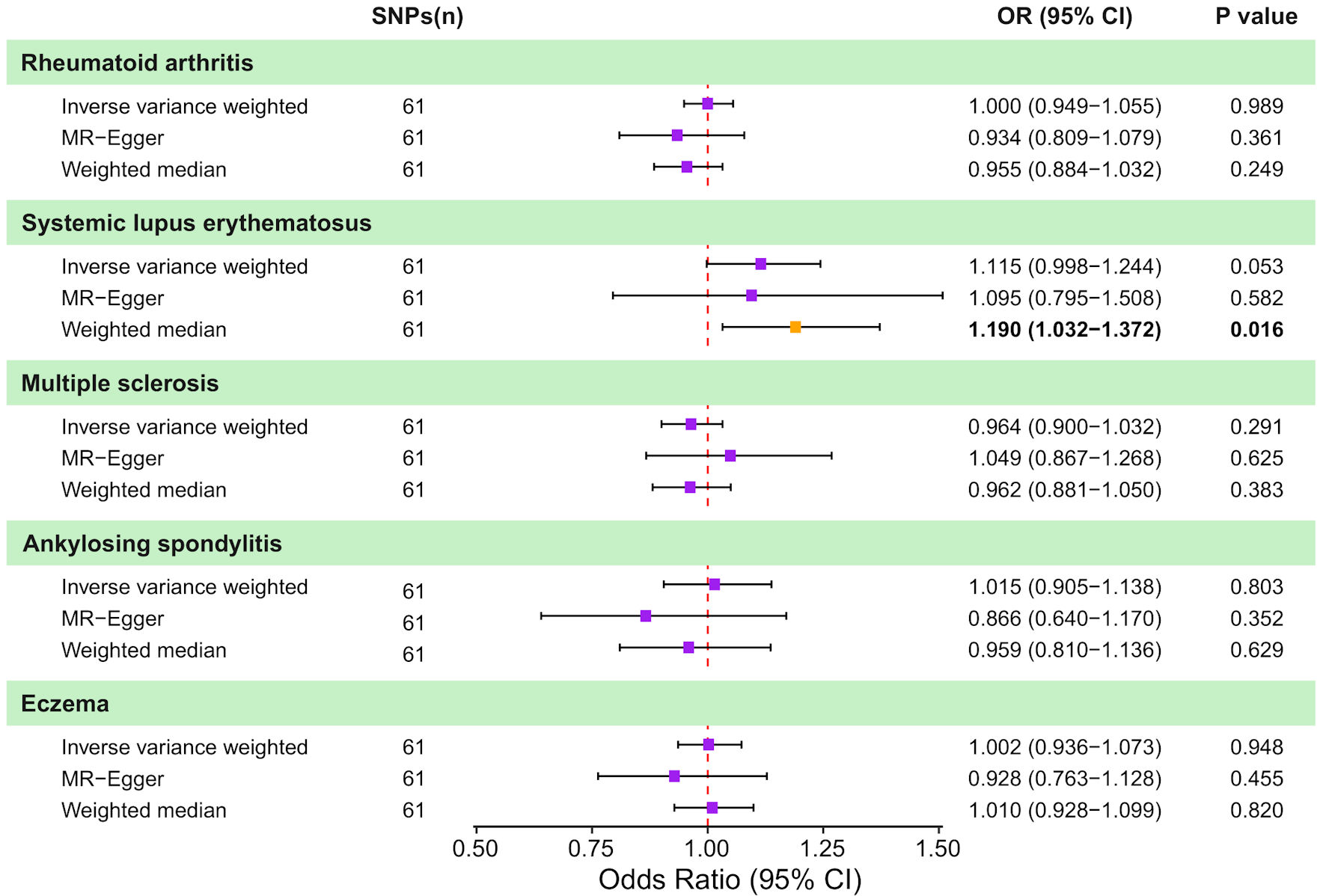
↓ Figure 4. Causal effects of genetically
predicted colorectal cancer (CRC) on autoimmune disease risk in two-sample Mendelian randomization (MR)
analyses. Forest plot shows the odds ratio (OR) and 95% confidence interval (CI) for the genetically
predicted liability of five autoimmune diseases to CRC. For each outcome, estimates are presented for
the inverse-variance weighted (IVW) method, MR-Egger regression, and the weighted median approach,
together with the corresponding number of instrumental SNPs and P values. Squares represent estimates
and horizontal lines indicate 95% CIs; the vertical dashed line denotes the null effect (OR = 1.0). SNP:
single nucleotide polymorphism.
↓ Figure 5. Integrated summary of Mendelian
randomization (MR) estimates and sensitivity analyses for colorectal cancer and autoimmune diseases. The
heatmap summarizes the strength of evidence from three MR methods (IVW, MR-Egger, and weighted median)
for testing whether genetic liability to colorectal cancer (CRC) is causally associated with risk of
eight autoimmune diseases (RA, SLE, CD, asthma, MS, gout, AS, and eczema). Red boxes indicate a
statistically significant causal association (P < 0.05), whereas blue boxes represent a lack of
evidence for a causal relationship. The upper panel summarizes sensitivity analyses, with green dots
indicating no evidence and red dots indicating the presence of outliers, horizontal pleiotropy, or
heterogeneity. Slashes denote an insufficient number of instrumental single-nucleotide polymorphisms
(SNPs) to compute MR estimates for specific analyses. RA: rheumatoid arthritis; SLE: systemic lupus
erythematosus; AS: ankylosing spondylitis; CD: celiac disease; IVW: inverse-variance weighted.
Table
↓ Table 1. Characteristics of the GWAS on Autoimmune Diseases and
Colorectal Cancer Used for MR Analyses
|
Variables |
PubMed identification |
Year
|
No.
of cases |
No.
of control participants |
Population |
Sex
|
SNPs
|
Sample size |
| aThe CRC data consist of Asian and European ancestry, from which European
ancestry data were extracted for subsequent analysis to ensure uniformity of the sample. GWAS:
genome-wide association study; MR: Mendelian randomization; SNPs: single-nucleotide polymorphisms.
|
| Rheumatoid arthritis [22] |
23143596 |
2012 |
13,838 |
33,742 |
European |
Males and females |
112,654 |
47,580 |
| Systemic lupus erythematosus [23] |
26502338 |
2015 |
5,201 |
9,066 |
European |
Males and females |
7,071,163 |
14,267 |
| Celiac disease [24] |
20190752 |
2010 |
4,533 |
10,750 |
European |
Males and females |
518,292 |
15,283 |
| Asthma [25] |
29273806 |
2018 |
19,954 |
107,715 |
European |
Males and females |
1,999,262 |
127,669 |
| Multiple sclerosis [26] |
31604244 |
2019 |
47,429 |
68,374 |
European |
Males and females |
6,304,359 |
115,803 |
| Gout [27] |
23263486 |
2013 |
2,115 |
67,259 |
European |
Males and females |
5,057,528 |
69,374 |
| Ankylosing spondylitis [28] |
/ |
2021 |
1,462 |
164,682 |
European |
Males and females |
16,380,022 |
166,144 |
| Eczema [29] |
26482879 |
2015 |
10,788 |
30,047 |
European |
Males and females |
11,059,641 |
40,835 |
| Colorectal cancera [30] |
36539618 |
2023 |
78,473 |
107,143 |
European (73%) and Asian (27%) |
Males and females |
11,738,639 |
185,616 |




