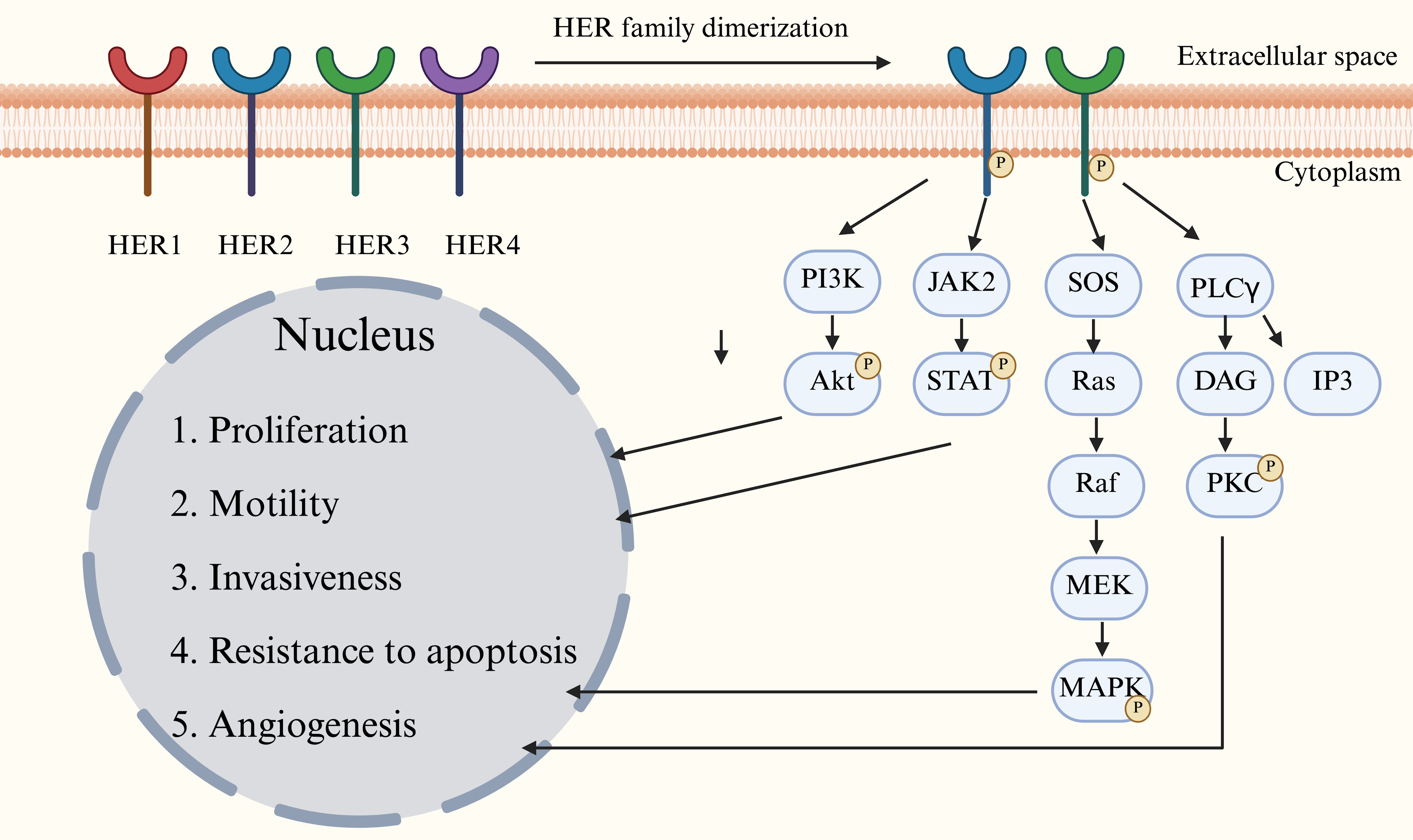
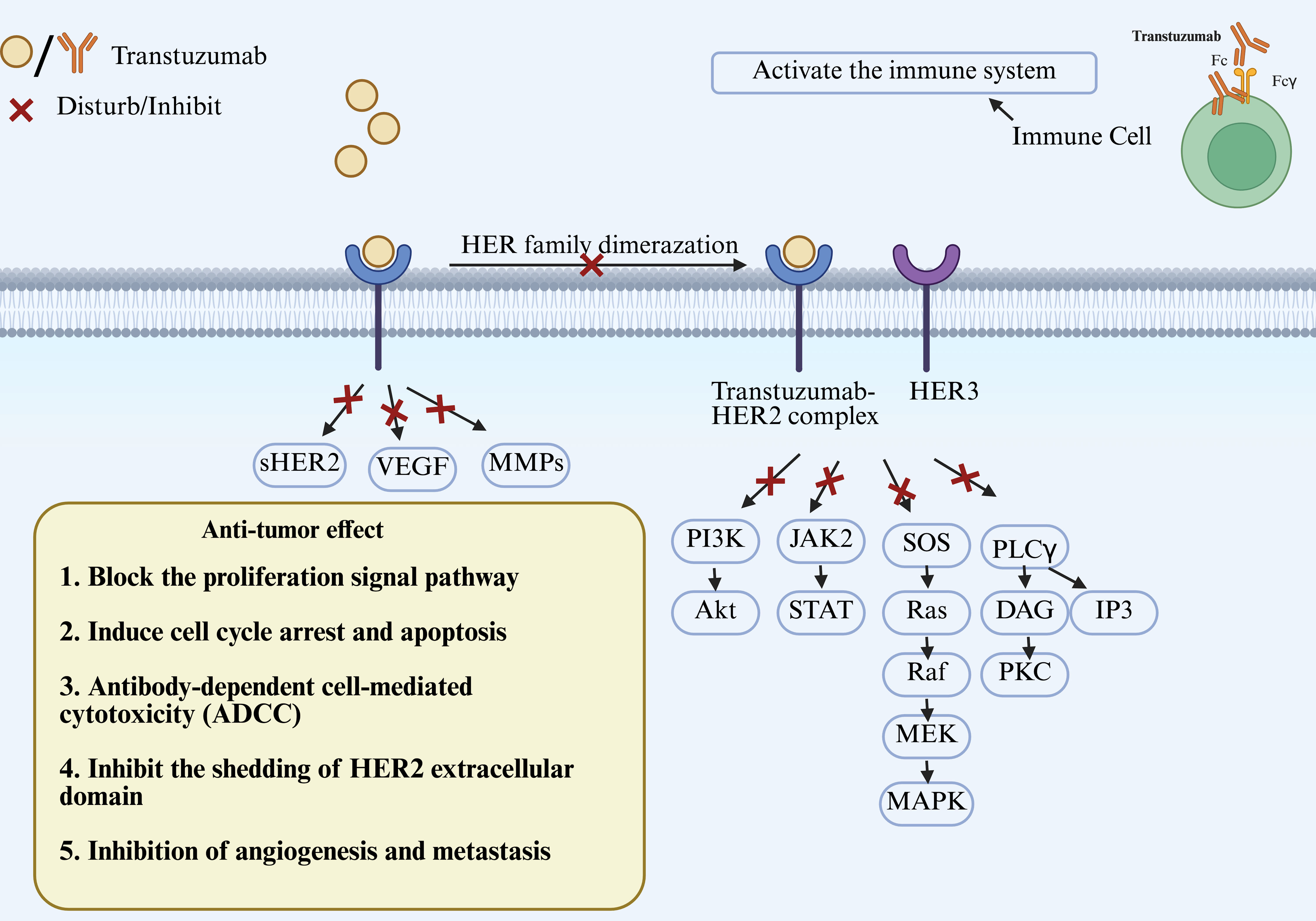
| DESTINY-PanTumor02 |
II |
Patients with previously treated, locally advanced or metastatic
HER2-expressing (IHC 3+/2+) solid tumors (including endometrium, cervix, ovary, bladder, biliary
tract, pancreas and other cancers) |
T-DXd |
For all patients: confirmed ORR: 37.1% (95% CI, 31.3–43.2); median
PFS: 6.9 months (95% CI, 5.6–8.0); median OS: 13.4 months (95% CI, 11.9–15.5) |
[54] |
|
|
|
|
For centrally confirmed IHC 3+ patients: confirmed ORR: 61.3% (95% CI,
49.4–72.4); median PFS: 11.9 months (95% CI, 8.2–13.0); median OS: 21.1 months (95%
CI, 15.3–29.6) |
|
| DESTINY-Breast04 |
III |
Patients with prior chemotherapy, unresectable or metastatic HER2-low
expression (IHC 1+ or IHC 2+/ISH–) breast cancer |
T-DXd vs. chemotherapy of doctor’s choice (TPC) |
Median PFS: 10.1 vs. 5.4 months (T-DXd vs. TPC); median OS 23.9 vs. 17.5
months (T-DXd vs. TPC) |
[55] |
| DESTINY-Breast06 |
III |
Patients with HR-positive, HER2-low, or very low-expressing mBC who have
progressed after at least first-line endocrine therapy and have not received metastatic chemotherapy
|
T-DXd vs. chemotherapy of doctor’s choice (TPC:
capecitabine/albumin paclitaxel/paclitaxel) |
For HER2-low expression population: median PFS: 13.2 vs. 8.1 months (T-DXd
vs. TPC). |
[56] |
|
|
|
|
For ITT population, including very low HER2 expression): median PFS: 13.2
vs. 8.1 months. |
|
| DESTINY-Gastric02 |
II |
Patients with HER2-positive advanced gastric or gastroesophageal junction
cancer with disease progression on or after a trastuzumab-containing regimen |
T-DXd |
Confirmed ORR: 41·8% (95% CI, 30·8–53·4); median
PFS: 5.6 months (95% CI, 4.2–8.3); median OS: 12.1 months (95% CI, 9.4–15.4); median
DoR: 8.1 months (95% CI, 5.9–NE); DCR: 81.0% (64/79; 95% CI, 70.6–89.0); median TTR:
1.4 months (95% CI, 1.4–2.7); CR: 5.1%; PR: 36.7%. |
[57] |
|
|
|
|
|
|
| DAISY |
II |
Patients with mBC stratified by HER2 expression level into three cohorts:
HER2-overexpressing, HER2-low, and HER2-non-expressing |
T-DXd |
Primary endpoint (confirmed ORR): cohort 1, 70.6% (95%, CI
58.3–81); cohort 2, 37.5% (95% CI, 26.4–49.7); cohort 3, 29.7% (95% CI,
15.9–47). |
[58] |
|
|
|
|
Secondary endpoints (median PFS): cohort 1, 11.1 months (95% CI,
8.5–14.4); cohort 2, 6.7 months (95% CI, 4.4–8.3); cohort 3, 4.2 months (95% CI,
2.0–5.7) |
|
|
|
|
|
Median OS (not fully mature at data cutoff): cohort 1 and cohort 2, not
reached; cohort 3, 11.6 months. |
|
|
|
|
|
CBR: cohort 1, 85.3%; cohort 2, 56.9%; cohort 3, 35.1%. |
|
|
|
|
|
DoR: cohort 1, 9.7 months; cohort 2, 7.6 months; cohort 3, 6.8 months.
|
|