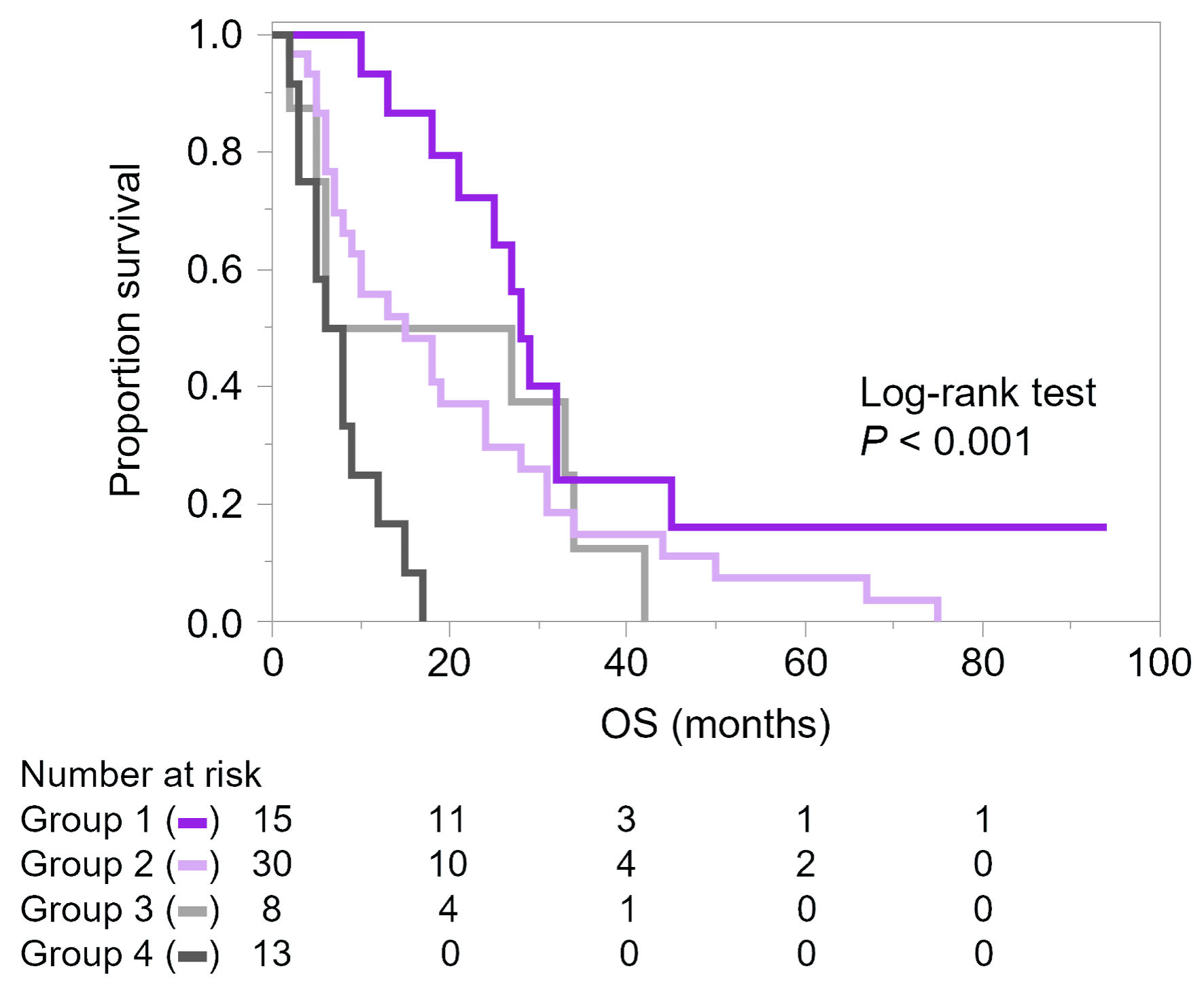
↓ Figure 1. Flowchart of eligible patients.
Patient selection criteria for the current study are shown. TNBC: triple-negative breast cancer.

| World Journal of Oncology, ISSN 1920-4531 print, 1920-454X online, Open Access |
| Article copyright, the authors; Journal compilation copyright, World J Oncol and Elmer Press Inc |
| Journal website https://wjon.elmerpub.com |
Original Article
Volume 17, Number 2, April 2026, pages 268-276
Clinical Outcomes and Prognostic Factors in Metastatic Triple-Negative Breast Cancer: A Real-World Data Analysis
Figures

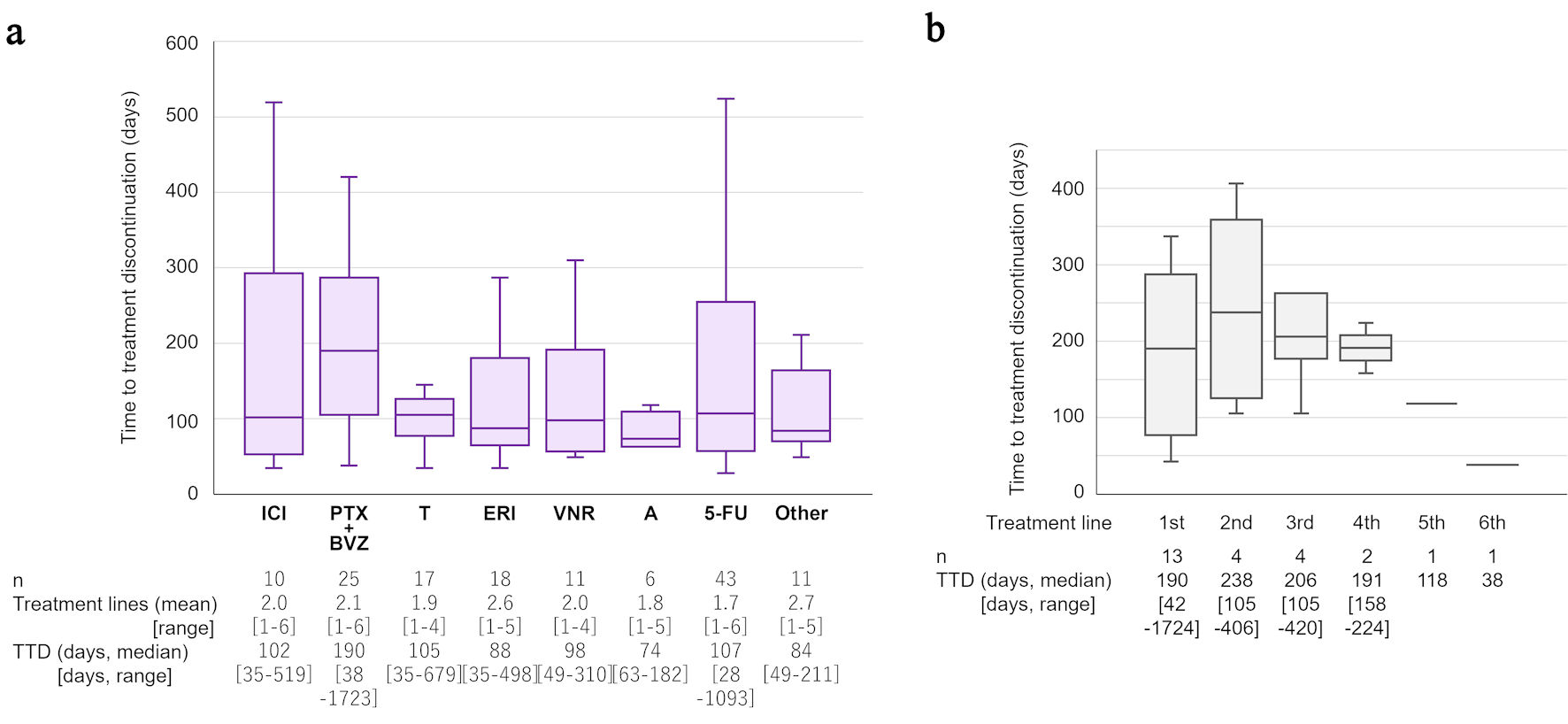
Tables
| Variables | Total (n = 96) | Treated group (n = 66) | Non-treated group (n = 30) | P value |
|---|---|---|---|---|
| aAt the time of diagnosis for distant metastasis. bIncluding neoadjuvant chemotherapy. IDC: invasive ductal carcinoma; NST: no special type; CT: chemotherapy; DFI: disease-free interval. | ||||
| Agea (mean, year) (range) | 57 (26–84) | 54 (26–84) | 59 (29–84) | 0.068 |
| Histology | ||||
| IDC (NST) | 86 (90%) | 60 (90%) | 26 (87%) | 0.528 |
| Others | 10 (10%) | 6 (10%) | 4 (13%) | |
| pT (mean, mm) (range) | 36 (0–125) | 37 (0–125) | 34 (0–115) | 0.721 |
| pN | ||||
| Positive | 53 (55%) | 38 (58%) | 15 (50%) | 0.489 |
| Negative | 43 (45%) | 28 (42%) | 15 (50%) | |
| Tumor grade | ||||
| High | 47 (49%) | 31 (47%) | 16 (53%) | 0.579 |
| Low/intermediate | 42 (44%) | 30 (45%) | 12 (40%) | |
| Not documented | 7 (7%) | 5 (8%) | 2 (7%) | |
| Adjuvant CTb | ||||
| Yes | 86 (90%) | 60 (90%) | 26 (87%) | 0.528 |
| No | 10 (10%) | 6 (10%) | 4 (13%) | |
| DFI (median, month) (range) | 11 (0–100) | 10 (0–70) | 12 (1–100) | 0.333 |
| Triger for detecting mTNBC | ||||
| Some symptoms | 35 (36%) | 17 (26%) | 18 (60%) | 0.005 |
| Routine examinations | 36 (38%) | 29 (44%) | 7 (23%) | |
| Not documented | 25 (26%) | 20 (30%) | 5 (17%) | |
| Performance status | ||||
| 0/1 | 83 (86%) | 66 (100%) | 17 (57%) | < 0.001 |
| 2 | 5 (5%) | 0 (0%) | 5 (17%) | |
| 3/4 | 8 (8%) | 0 (0%) | 8 (27%) | |
| Number of metastatic sitesa (mean) (range) | 1.4 (1–4) | 1.4 (1–4) | 1.4 (1–3) | 0.970 |
| Visceral metastasis | ||||
| Yes | 59 (61%) | 41 (62%) | 18 (60%) | 0.843 |
| No | 37 (39%) | 25 (38%) | 12 (40%) | |
| Metastatic site | ||||
| Bone | 18 (19%) | 15 (23%) | 3 (10%) | 0.139 |
| Liver | 24 (25%) | 14 (21%) | 10 (33%) | 0.204 |
| Lung | 40 (42%) | 28 (42%) | 12 (40%) | 0.823 |
| Brain | 15 (16%) | 4 (6%) | 11 (37%) | < 0.001 |
| Others | 39 (41%) | 32 (48%) | 7 (23%) | 0.020 |
| Variables | Univariate | Multivariate | ||||
|---|---|---|---|---|---|---|
| HR | 95% CI | P value | HR | 95% CI | P value | |
| aAt the time of diagnosis for distant metastasis. bThe range odds ratio. OS: overall survival; HR: hazard ratio; CI: confidence interval; NST: no special type; CT: chemotherapy; DFI: disease-free interval; ALC: absolute lymphocyte count; NLR: neutrophil-to-lymphocyte ratio; ICI: immune checkpoint inhibitor-based treatment; PTX: paclitaxel; BVZ: bevacizumab. | ||||||
| Agea | 0.33b | 0.1–0.9 | 0.023 | 0.41b | 0.1–1.8 | 0.240 |
| Histology, NST vs. others | 0.88 | 0.4–2.1 | 0.768 | |||
| High tumor grade, yes vs. no | 1.68 | 0.9–2.9 | 0.062 | 1.03 | 0.5–2.0 | 0.925 |
| HER2, low vs. null | 0.92 | 0.5–1.6 | 0.762 | |||
| Adjuvant CT, yes vs. no | 1.98 | 0.8–4.6 | 0.119 | |||
| DFI (months) | 0.46b | 0.1–1.5 | 0.210 | 0.90b | 0.2–4.3 | 0.894 |
| Number of metastatic sitesa | 4.81b | 1.6–13.0 | 0.007 | 8.88b | 1.2–50.2 | 0.020 |
| Visceral metastasisa, yes vs. no | 1.25 | 0.7–2.2 | 0.422 | |||
| Metastatic site | ||||||
| Bone | 1.55 | 0.8–2.9 | 0.158 | |||
| Liver | 1.29 | 0.7–2.5 | 0.438 | |||
| Lungs | 0.93 | 0.6–1.6 | 0.794 | |||
| Brain | 3.66 | 1.3–10.5 | 0.016 | 0.74 | 0.1–4.3 | 0.739 |
| ALCa | 0.46b | 0.1–2.0 | 0.321 | |||
| NLRa | 1.35b | 0.5–3.4 | 0.544 | 1.14b | 0.4–3.3 | 0.811 |
| Administration of ICI, yes vs. no | 0.70 | 0.3–1.8 | 0.444 | |||
| Administration of PTX + BVZ, yes vs. no | 0.54 | 0.3–0.9 | 0.028 | 0.44 | 0.2–0.9 | 0.029 |