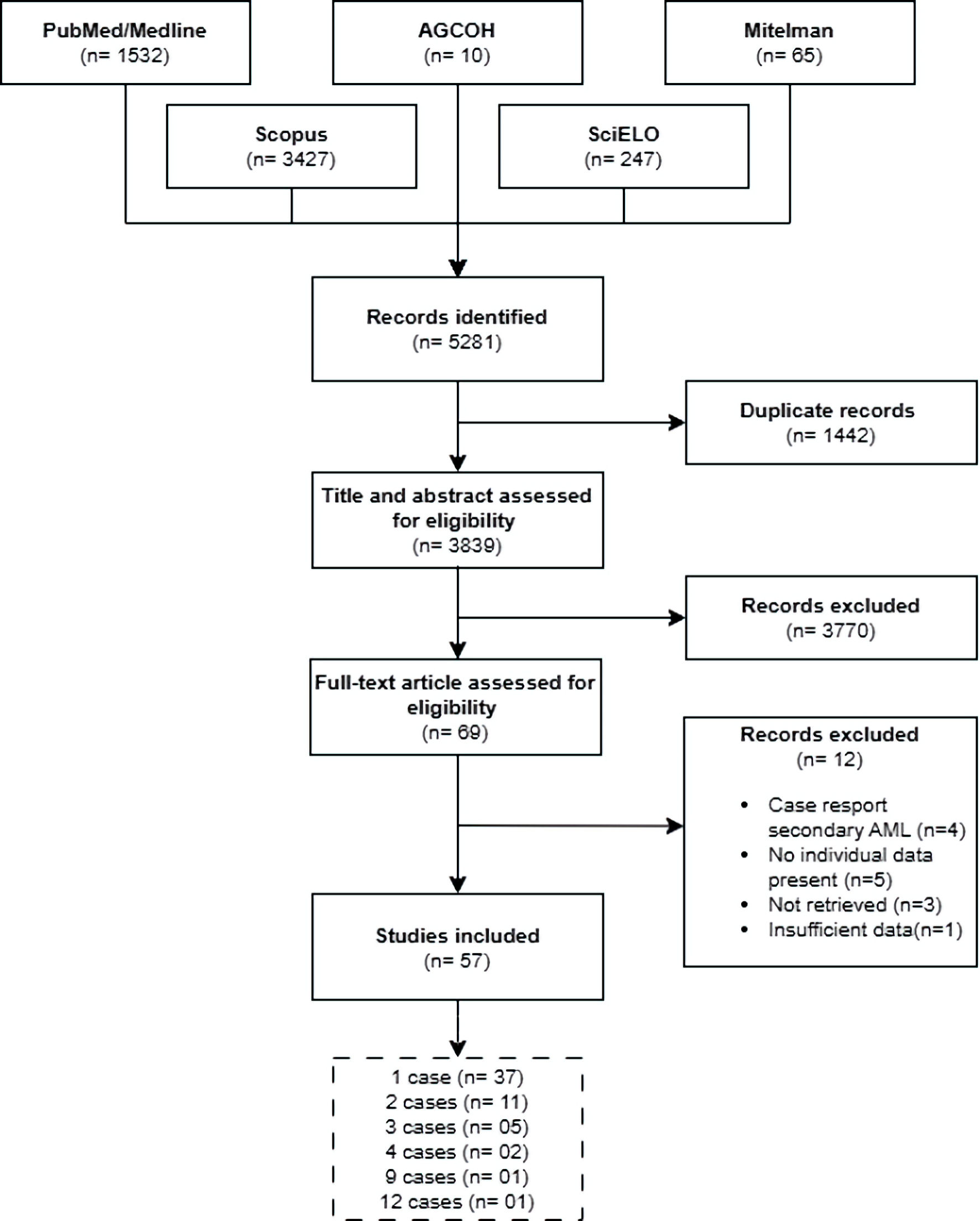
↓ Figure 1. Flowchart of article selection. AML:
acute myeloid leukemia; AGCOH: Atlas of Genetics and Cytogenetics in Oncology and Hematology.
| World Journal of Oncology, ISSN 1920-4531 print, 1920-454X online, Open Access |
| Article copyright, the authors; Journal compilation copyright, World J Oncol and Elmer Press Inc |
| Journal website https://wjon.elmerpub.com |
Original Article
Volume 17, Number 2, April 2026, pages 178-190
Characterization of Acute Myeloid Leukemia With t(16;21) Translocation: Cytogenetic, Molecular, and Immunophenotypic Findings
Figures

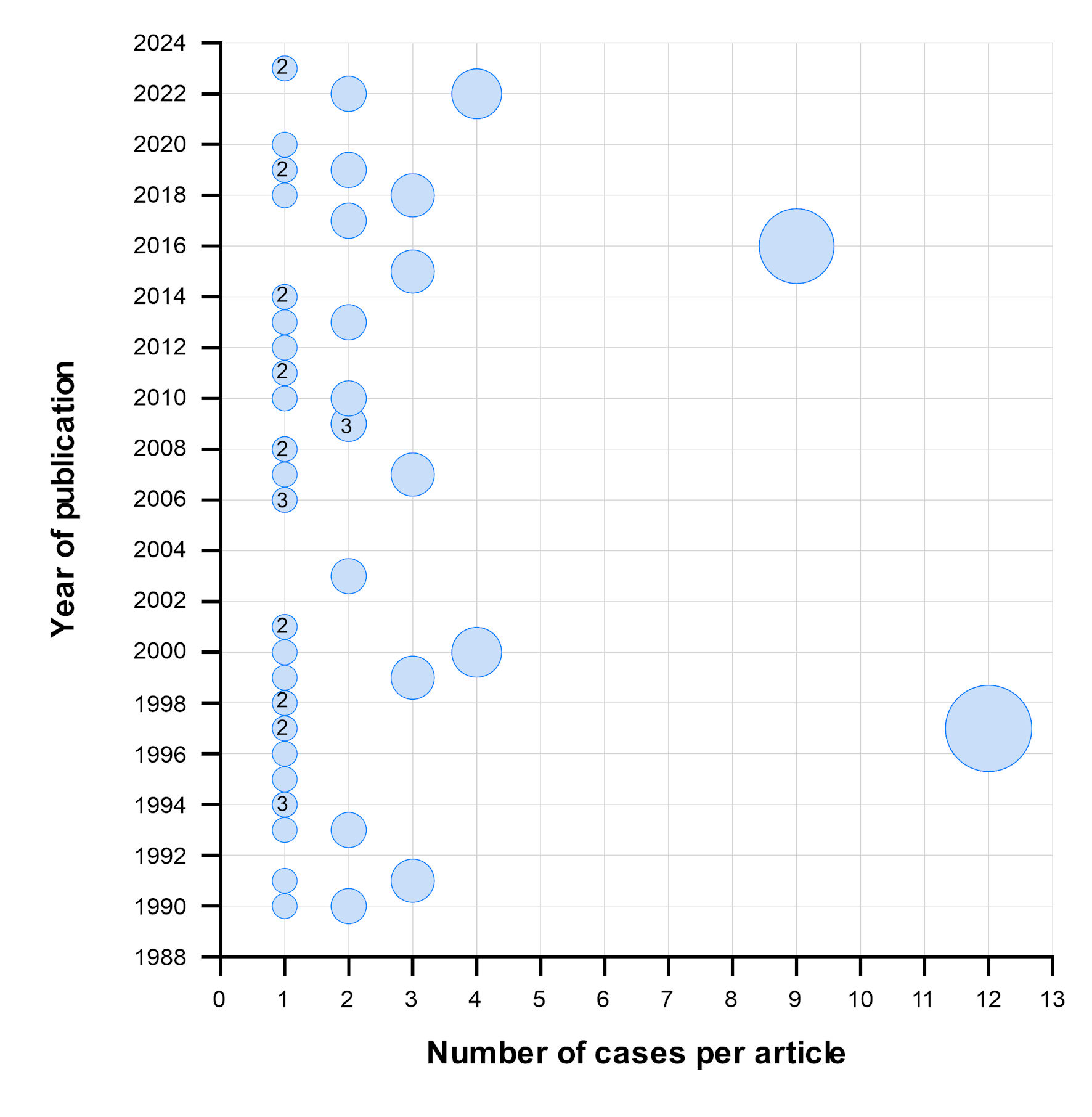
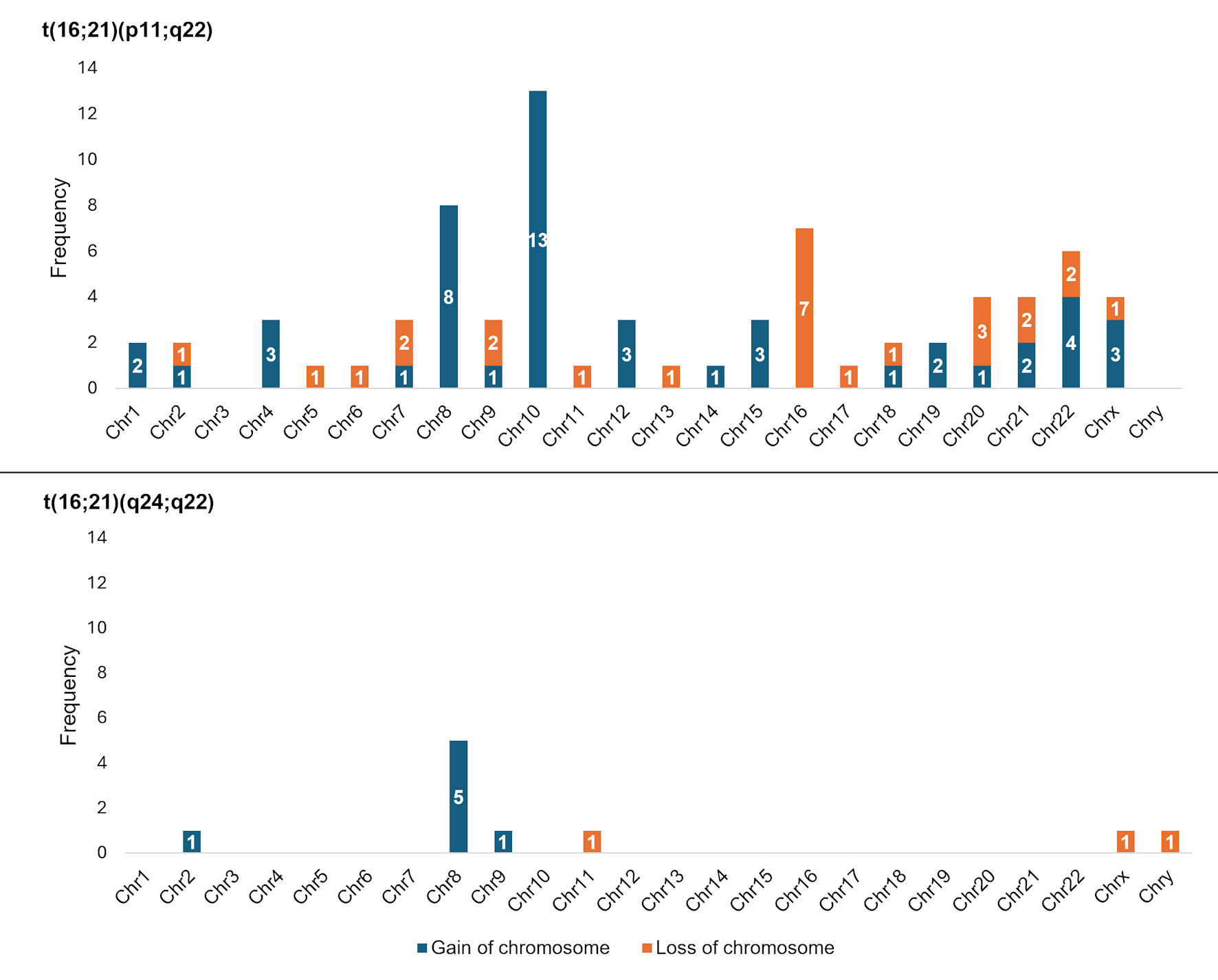
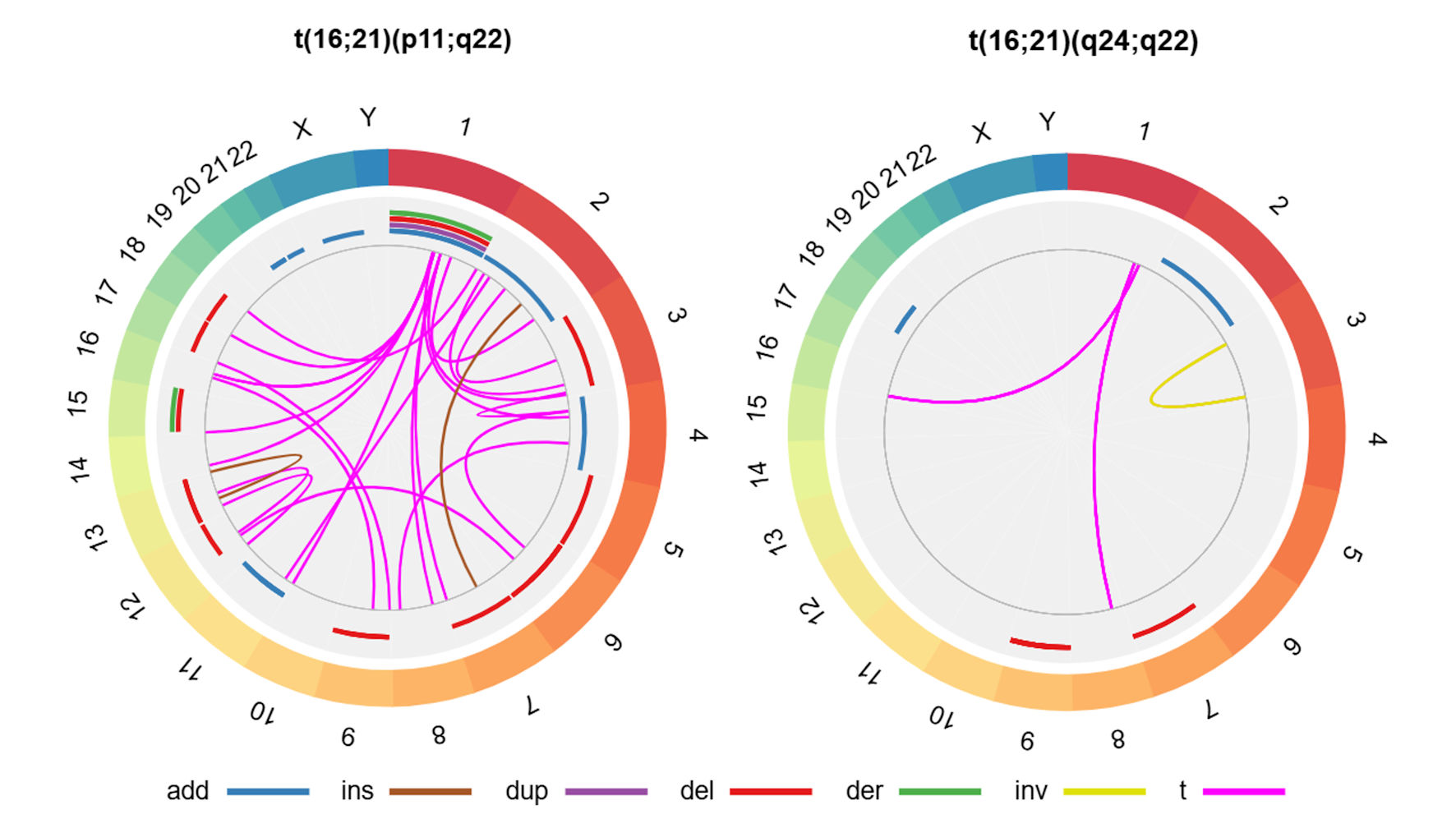
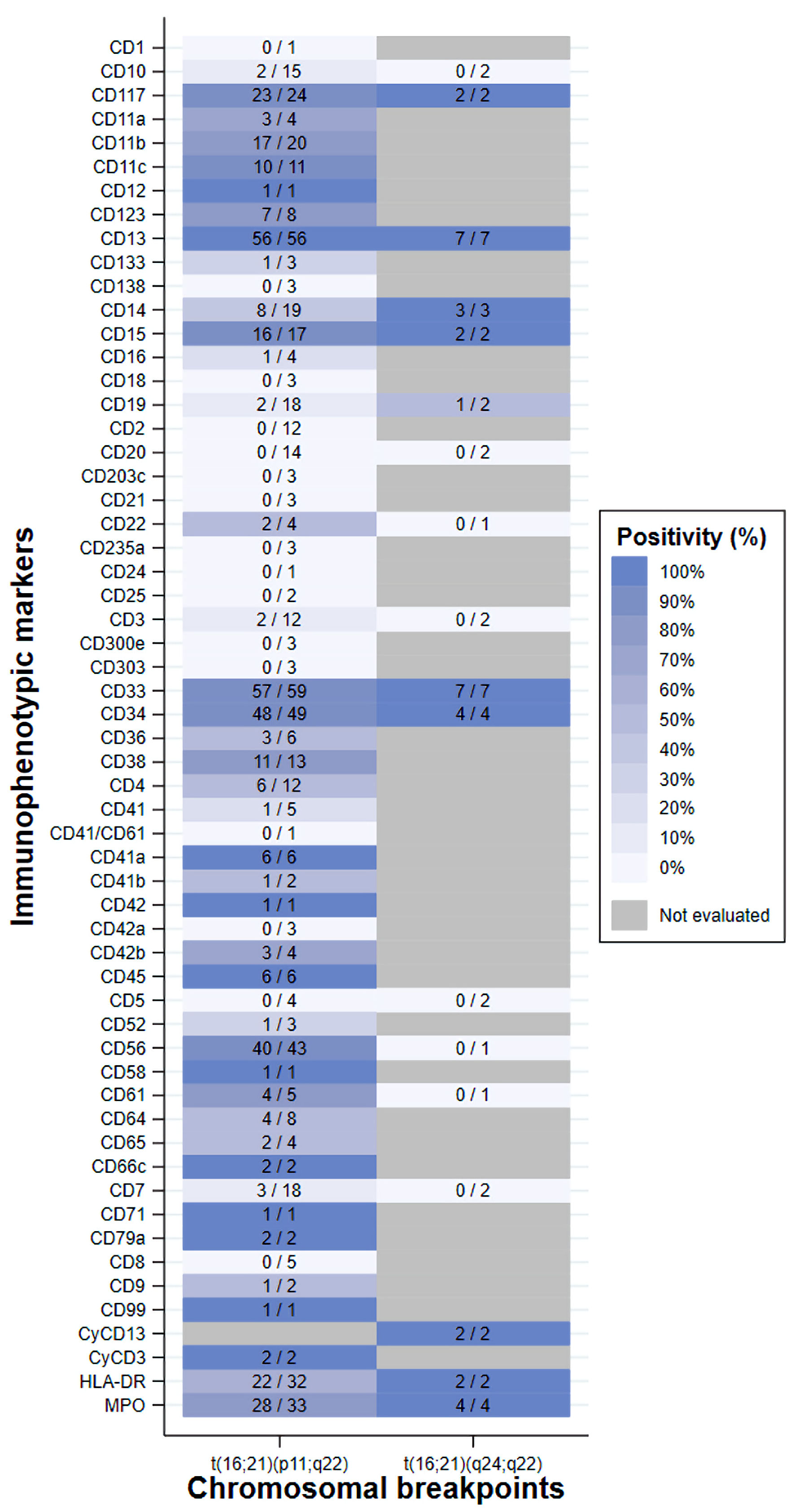
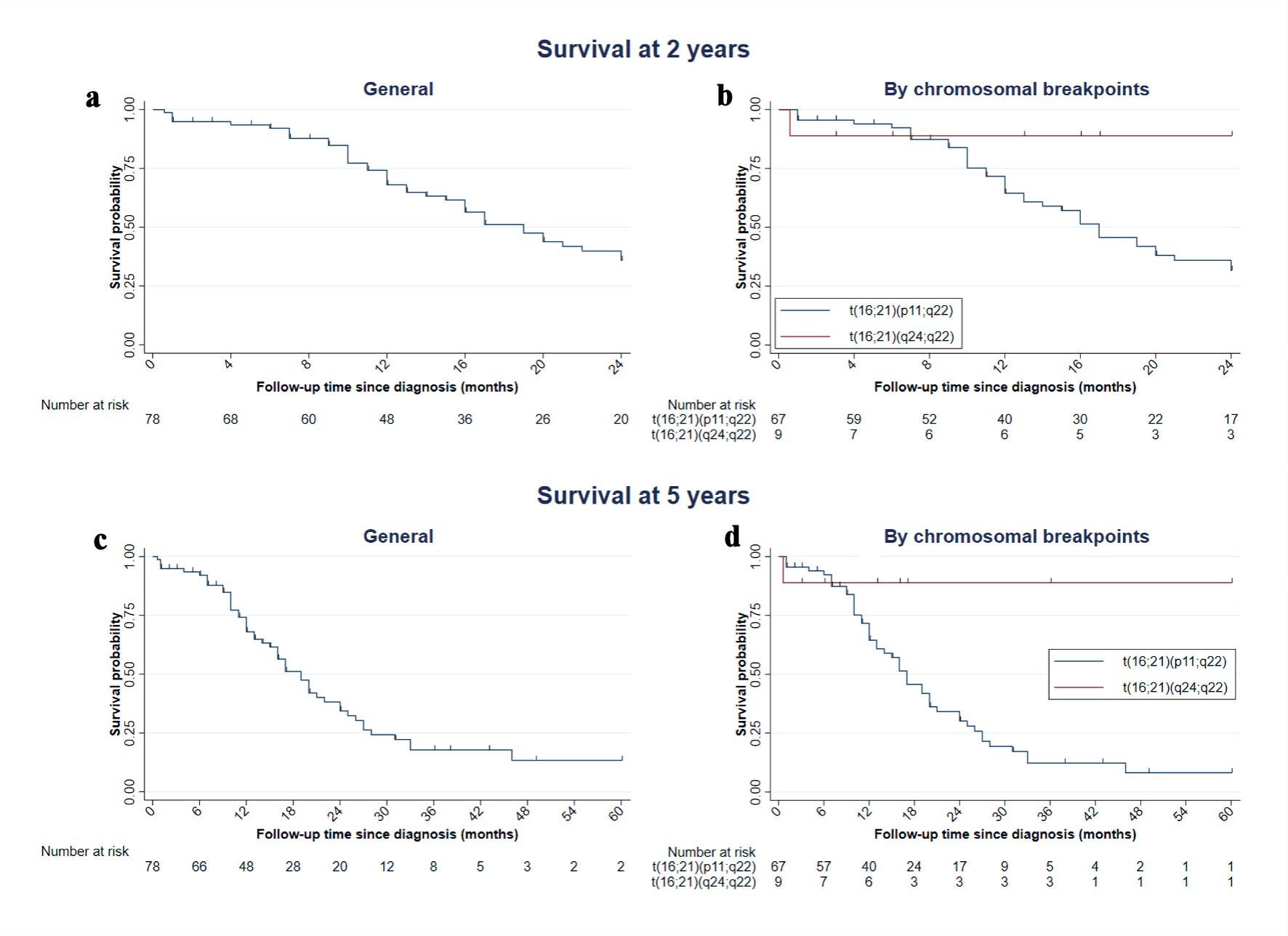
Table
| Characteristic | Overalla (n = 103), n (%) | t(16;21) breakpoints | P value | |
|---|---|---|---|---|
| (p11;q22) (n = 90), n (%) | (q24;q22) (n = 11), n (%) | |||
| aTwo additional cases were identified: the first case was in an adult, 46,XX, with t(16;21)(p11:q13) FAB-M5 who received chemotherapy plus transplantation and experienced relapse; the second case was in an adult, 46,XX, with t(16;21)(q21;p21) FAB-M5 with secondary structural chromosomal alterations who received chemotherapy plus transplantation and did not experience relapse. bMedian (IQR). cFisher’s exact test. dMann–Whitney U test. FAB: French–American–British; M5: acute monoblastic or monocytic leukemia; IQR: interquartile range; NA: not applicable; t(16;21): translocation between chromosomes 16 and 21. | ||||
| Sex (male) | 56 (54.4) | 50 (55.6) | 6 (54.5) | 1.000c |
| Age (years)b | 23 (9–39) | 24 (10.5–40.5) | 6 (4–71) | 0.250d |
| Age group | 0.009c | |||
| Pediatric (< 18 years) | 44 (42.7) | 37 (41.1) | 7 (63.6) | |
| Adult (18–59 years) | 49 (47.6) | 46 (51.1) | 1 (9.1) | |
| Older adult (≥ 60 years) | 8 (7.8) | 5 (5.6) | 3 (27.3) | |
| No information | 2 (1.9) | 2 (2.2) | 0 (0.0) | |
| FAB classification | 0.326c | |||
| M0: AML with minimal differentiation | 1 (1) | 1 (1.1) | 0 (0.0) | |
| M1: AML without maturation | 22 (21.4) | 20 (22.2) | 2 (18.2) | |
| M2: AML with maturation | 25 (24.3) | 21 (23.3) | 4 (36.4) | |
| M4: acute myelomonocytic leukemia | 16 (15.5) | 12 (13.3) | 4 (36.4) | |
| M5: acute monoblastic/monocytic leukemia | 22 (21.4) | 20 (22.2) | 0 (0.0) | |
| M6: acute erythroid leukemia | 1 (1) | 1 (1.1) | 0 (0.0) | |
| M7: acute megakaryoblastic leukemia | 8 (7.8) | 8 (8.9) | 0 (0.0) | |
| NOS: AML not otherwise specified | 1 (1) | 1 (1.1) | 0 (0.0) | |
| No information | 7 (6.8) | 6 (6.7) | 1 (9.1) | |
| Presence of numerical alterations (yes) | 39 (37.9) | 32 (35.6) | 7 (63.6) | 0.101c |
| Number of chromosomes | 0.379c | |||
| < 46 | 6 (5.8) | 6 (6.7) | 0 (0.0) | |
| 46 | 78 (75.7) | 69 (76.7) | 7 (63.6) | |
| > 46 | 17 (16.5) | 13 (14.4) | 4 (36.4) | |
| No information | 2 (1.9) | 2 (2.2) | 0 (0.0) | |
| Presence of structural alterations (yes) | 47 (45.6) | 40 (44.4) | 6 (54.5) | 0.542c |
| Primary molecular alterations | < 0.001c | |||
| RUNX1::RUNX1T3 | 8 (7.8) | NA | 8 (72.7) | |
| FUS::ERG | 56 (54.4) | 56 (62.2) | NA | |
| No information | 39 (37.9) | 34 (37.8) | 3 (27.3) | |
| Treatment | 0.002c | |||
| Chemotherapy | 53 (51.5) | 47 (52.2) | 6 (54.5) | |
| Chemotherapy + transplant | 30 (29.1) | 28 (31.1) | 0 (0.0) | |
| Other | 2 (1.9) | 0 (0.0) | 2 (18.2) | |
| No information | 18 (17.5) | 15 (16.7) | 3 (27.3) | |
| Relapse | 0.008c | |||
| Present | 51 (49.5) | 49 (54.4) | 1 (9.1) | |
| Absent | 16 (15.5) | 12 (13.3) | 3 (27.3) | |
| No information | 36 (35.0) | 29 (32.2) | 7 (63.6) | |