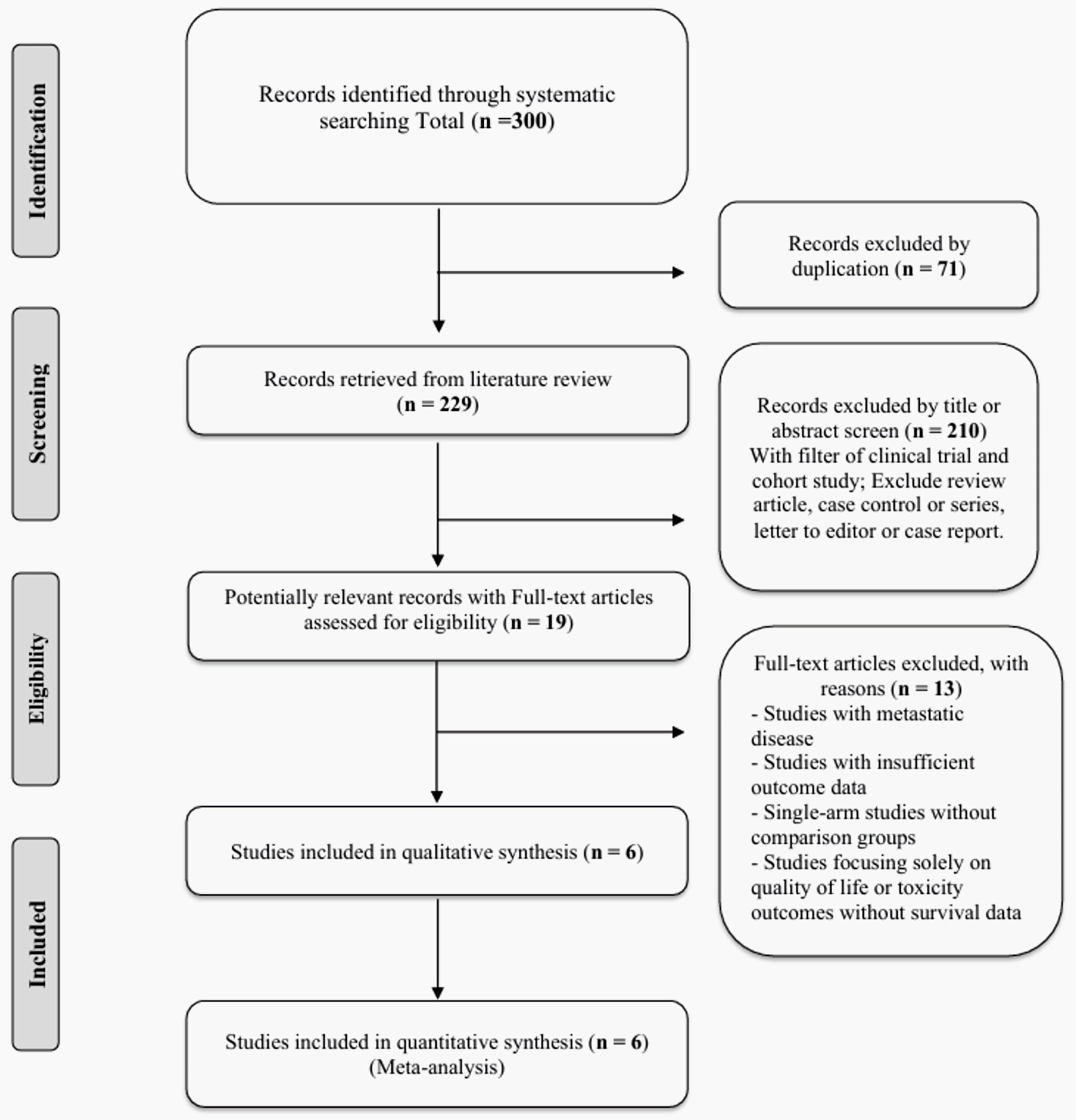
↓ Figure 1. PRISMA flow diagram for study
selection. PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
| World Journal of Oncology, ISSN 1920-4531 print, 1920-454X online, Open Access |
| Article copyright, the authors; Journal compilation copyright, World J Oncol and Elmer Press Inc |
| Journal website https://wjon.elmerpub.com |
Original Article
Volume 16, Number 6, December 2025, pages 609-620
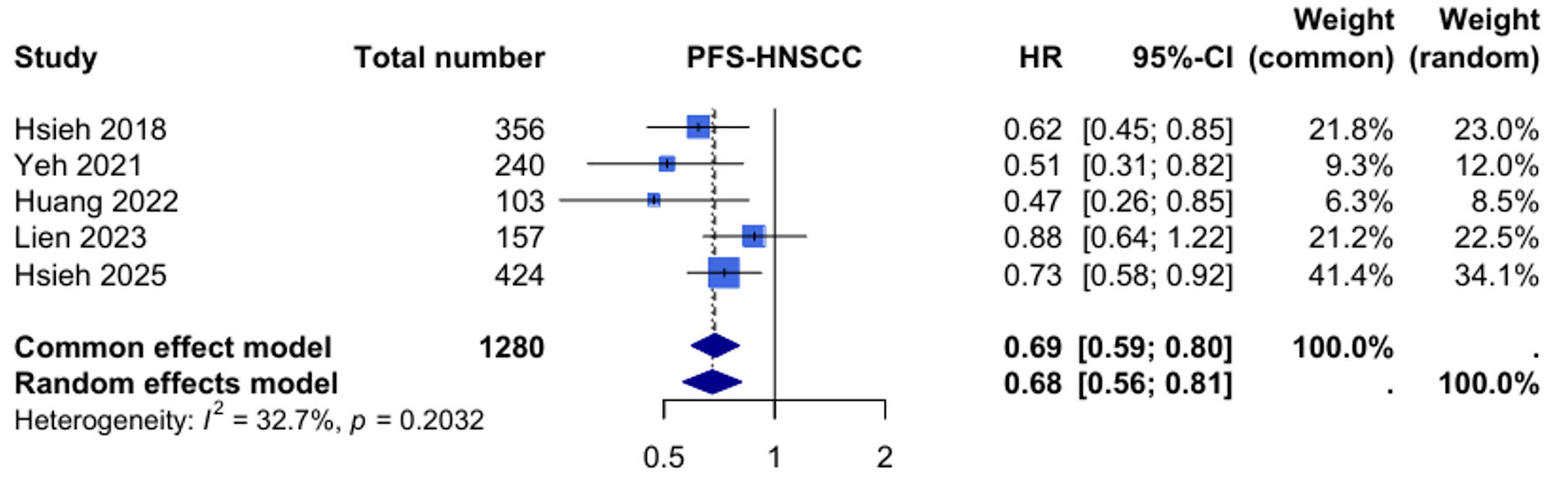
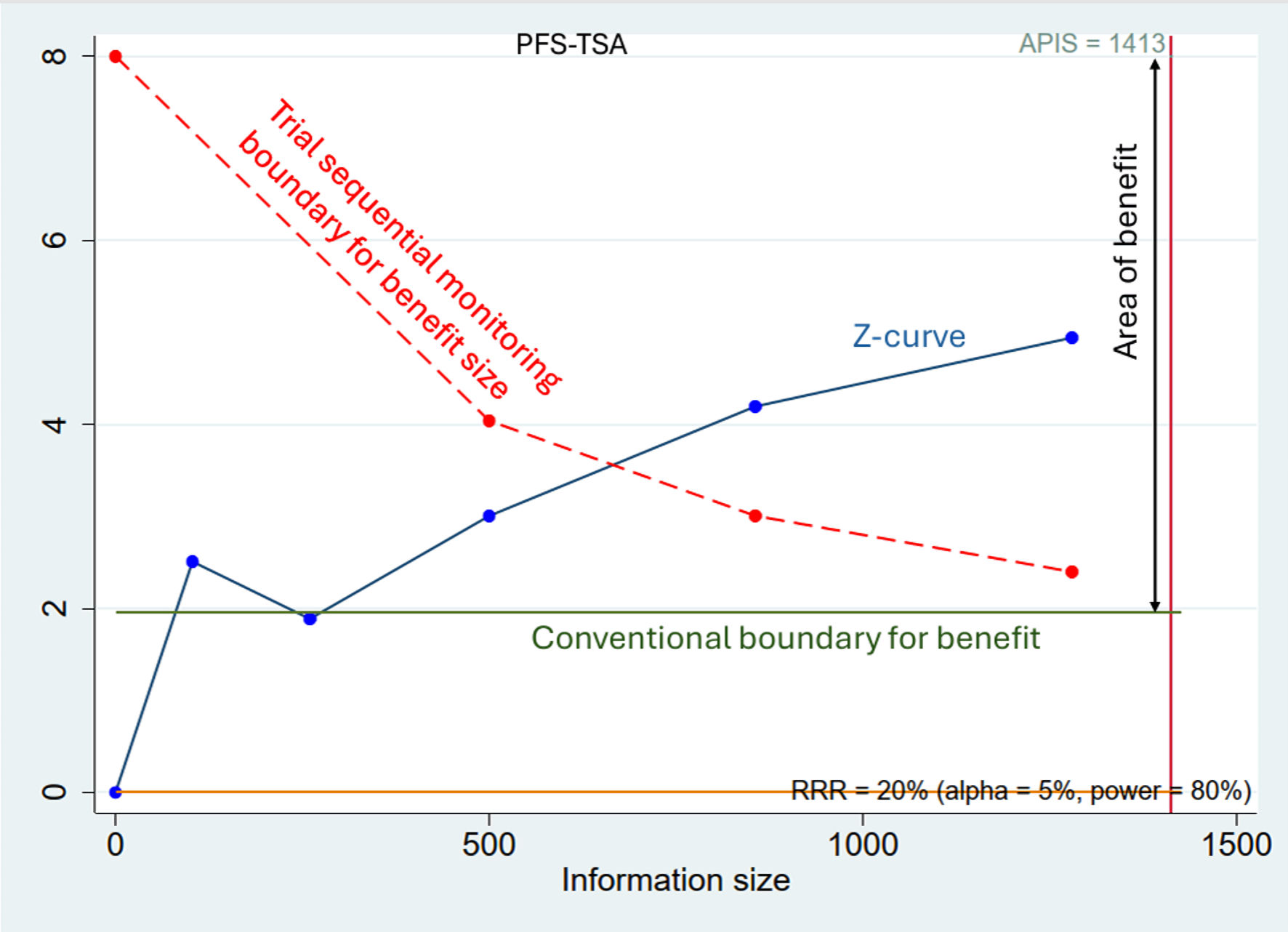
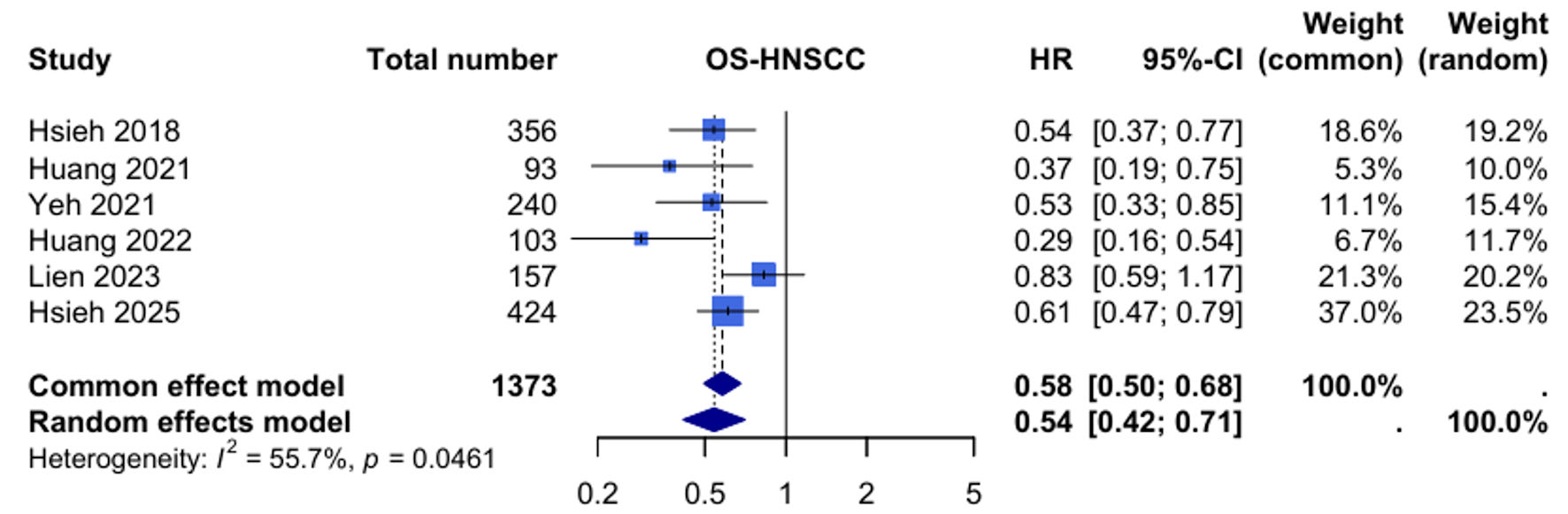
Efficacy of Tegafur-Uracil Maintenance Therapy in Non-Metastatic Head and Neck Squamous Cell Carcinoma: A Meta-Analysis With Trial Sequential Analysis
Figures

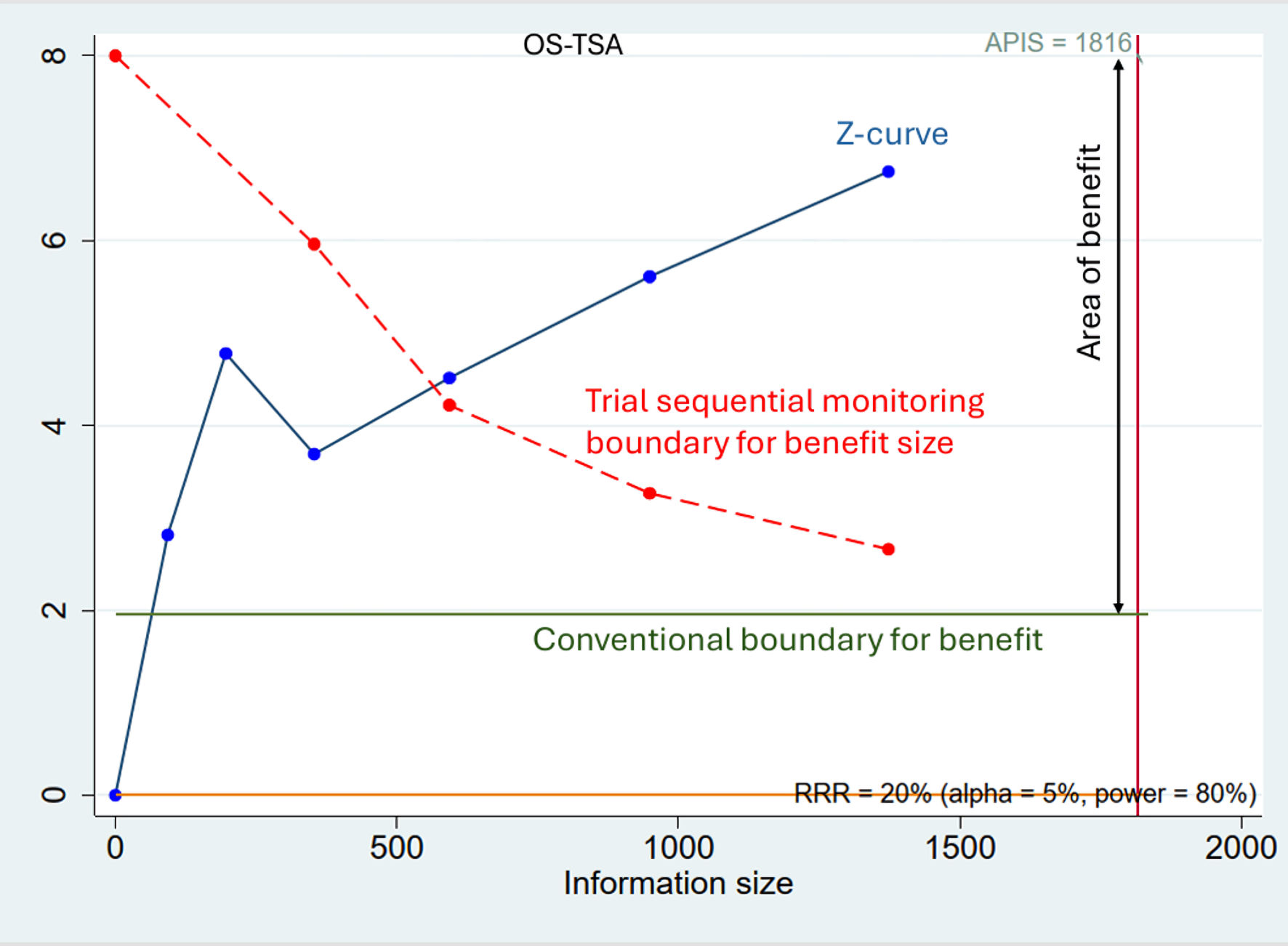
Tables
| Study (HNSCC) | Cancer type | Stage | Prior treatment | Treatment groups | Sample size |
|---|---|---|---|---|---|
| UFT: tegafur-uracil; HNSCC: head and neck squamous cell carcinoma; CCRT: concurrent chemoradiotherapy; OC: oral cavity; OPx: oropharynx; HPx: hypopharynx; Lx: larynx. | |||||
| Huang et al, 2021 [19] | OC | IVa and IVb | Surgical resection and CCRT with cisplatin-based regimen | UFT vs. non-UFT | 93 (51 UFT, 42 non-UFT) |
| Huang et al, 2022 [20] | OC | IVa and IVb | Curative surgical resection followed by adjuvant cisplatin-based CCRT | UFT vs. non-UFT | 103 (64 UFT, 39 non-UFT) |
| Lien et al, 2023 [21] | HPx | 25% III, 75% IVa-IVb | Total laryngopharyngectomy followed by adjuvant CRT | UFT vs. non-UFT in cortactin-positive and cortactin-negative patients | 157 total (53 cortactin+, 104 cortactin-) |
| Yeh et al, 2021 [22] | OC, OPx, HPx | 21% III, 79% IVa-IVb | Surgical resection, CCRT or chemotherapy | UFT vs. non-UFT | 240 (96 UFT, 144 non-UFT) |
| Hsieh et al, 2018 [9] | OC | 13% III, 87% IV | Curative surgery and postoperative chemoradiotherapy | UFT vs. non-UFT | 356 (114 UFT, 242 non-UFT) |
| Hsieh et al, 2025 [23] | OC, OPx, HPx, Lx | 9% stage I-II, 91% stage III-IVB | Definitive chemoradiotherapy without surgical intervention | UFT vs. non-UFT | 424 (212 UFT, 212 non-UFT) |
| Study (HNSCC) | Mean age (years) | Gender (male %) | ECOG PS (0 - 1) % | T classification | N classification |
|---|---|---|---|---|---|
| UFT: tegafur-uracil; HNSCC: head and neck squamous cell carcinoma; ECOG PS: Eastern Cooperative Oncology Group performance status. | |||||
| Huang et al, 2021 [19] | UFT: 51.61 | UFT: 90.2% | UFT: 92.0% | T3-T4: UFT 72.55% | N2-N3: UFT 60.78%, non-UFT 57.14% |
| Non-UFT: 57.10 | Non-UFT: 88.1% | Non-UFT: 66.66% | Non-UFT 73.81% | ||
| Huang et al, 2022 [20] | UFT: 49.8 ± 8.8 | UFT: 90.6% | 100% (all ECOG PS 1) | T4: UFT 46.9% | N3B: UFT 82.8%, non-UFT 92.3% |
| Non-UFT: 49.9 ± 11.5 | Non-UFT: 92.3% | Non-UFT 61.5% | |||
| Lien et al, 2023 [21] | Median: 54 | 98% (both groups) | Not reported | pT3-4: cortactin+ 70%, cortactin- 65% | pN3: cortactin+ 68%, cortactin- 51% |
| Yeh et al, 2021 [22] | UFT: 56 ± 10 | UFT: 97.9% | Not reported | Not reported | Not reported |
| Non-UFT: 54 ± 10 | Non-UFT: 97.2% | ||||
| Hsieh et al, 2018 [9] | UFT: 52 ± 10 | UFT: 98% | Not reported | Not reported | Not reported |
| Non-UFT: 54 ± 11 | Non-UFT: 93% | ||||
| Hsieh et al, 2025 [23] | UFT: 42% > 60 years | UFT: 95% | Not reported | T1-T2: UFT 39%, non-UFT 41%; | N0-N1: UFT 37%, non-UFT 30% |
| Non-UFT: 36% > 60 years | Non-UFT: 92% | T3-T4: UFT 61%, non-UFT 59% | N2-N3: UFT 63%, non-UFT 70% | ||